Study on Nitrile Oxide for Low-Temperature Curing of Liquid Polybutadiene
- PMID: 35591729
- PMCID: PMC9101573
- DOI: 10.3390/ma15093396
Study on Nitrile Oxide for Low-Temperature Curing of Liquid Polybutadiene
Abstract
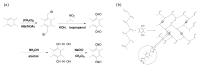
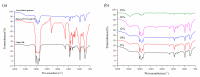
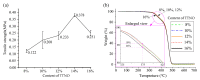
As a significant component of composite solid propellants, the cross-link alkenyl polymers need to cure at high temperatures and the current isocyanate curing systems are highly humidity sensitive. This paper presented a low-temperature curing method for a cross-linked polymer (polybutadiene) with stable wettability by using cycloaddition of the nitrile oxide of tetramethyl-terephthalobisnitrile oxide (TTNO) and the C=C group of liquid polybutadiene (PB). The TTNO was synthesized in four steps from 1,2,4,5-tetramethylbenzene and evaluated as a low-temperature hardener for curing liquid PB. To characterize the reaction ability of TTNO at 25 °C, the cross-linked rubber materials of various contents (8%, 10%, 12%, 14%, 16%) of curing agent TTNO were prepared. The feasibility of the curing method can be proved by the disappearance of the absorption peak of the nitrile oxide group (2300 cm-1) by FT-IR analysis. Contact angle, TG-DTA and tensile-test experiments were conducted to characterize the wettability, thermo-stability and mechanical properties of the obtained cross-linked rubber materials, respectively. The results showed that the curing agent TTNO could cure PB at room temperature. With the growing content of the curing agent TTNO, the tensile strength of the obtained cross-linked rubber material increased by 260% and the contact angle increased from 75.29° to 89.44°. Moreover, the thermo-stability performances of the cross-linked rubber materials have proved to be very stable, even at a temperature of 300 °C, by TGA analysis.
Keywords: cross-linked rubber material; low-temperature curing; tetramethyl-terephthalobisnitrile oxide.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
The dynamic interaction of water with four dental impression materials during cure.J Prosthodont. 2009 Jun;18(4):292-300. doi: 10.1111/j.1532-849X.2008.00423.x. Epub 2009 Jan 30. J Prosthodont. 2009. PMID: 19210607
-
Mechanical and Dynamic Behavior of an Elastic Rubber Layer with Recycled Styrene-Butadiene Rubber Granules.Polymers (Basel). 2020 Dec 17;12(12):3022. doi: 10.3390/polym12123022. Polymers (Basel). 2020. PMID: 33348745 Free PMC article.
-
Effect of Organoclay Addition on Rheological, Thermal, and Mechanical Properties of Nitrile Rubber/Phenolic Resin Blend.Polymers (Basel). 2022 Apr 3;14(7):1463. doi: 10.3390/polym14071463. Polymers (Basel). 2022. PMID: 35406336 Free PMC article.
-
Characterization of nanostructured epoxy networks modified with isocyanate-terminated liquid polybutadiene.J Colloid Interface Sci. 2011 Jun 15;358(2):338-46. doi: 10.1016/j.jcis.2011.03.030. Epub 2011 Mar 15. J Colloid Interface Sci. 2011. PMID: 21463862
-
Improving Thermo-Oxidative Stability of Nitrile Rubber Composites by Functional Graphene Oxide.Materials (Basel). 2018 May 30;11(6):921. doi: 10.3390/ma11060921. Materials (Basel). 2018. PMID: 29848944 Free PMC article.
References
-
- Khan M.A.S., Dey A., Athar J., Sikder A.K. Calculation of enthalpies of formation and band gaps of polymeric binders. RSC Adv. 2014;4:32840–32846. doi: 10.1039/C4RA02847C. - DOI
-
- Cohen A., Yan Q.-L., Shlomovich A., Aizikovich A., Petrutik N., Gozin M. Novel nitrogen-rich energetic macromolecules based on 3,6-dihydrazinyl-1,2,4,5-tetrazine. RSC Adv. 2015;5:106971–106980. doi: 10.1039/C5RA21345B. - DOI
-
- Cauty F., Fabignon Y., Erades C. New active binder-based propellants: A comparison with classical composite AP/HTPB propellants. Int. J. Energetic Mater. Chem. Propuls. 2013;12:1–13. doi: 10.1615/IntJEnergeticMaterialsChemProp.2013005271. - DOI
-
- Pant C.S., Santosh M.S., Banerjee S., Khanna P.K. Single Step Synthesis of Nitro-Functionalized Hydroxyl-Terminated Polybutadiene. Propell. Explos. Pyrotech. 2013;38:748–753. doi: 10.1002/prep.201300031. - DOI
-
- Reshmi S., Arunan E., Nair R. Azide and alkyne terminated polybutadiene binders: Synthesis, cross-linking, and propellant studies. Ind. Eng. Chem. Res. 2014;53:16612–16620. doi: 10.1021/ie502035u. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

