A Toll-like Receptor-Activating, Self-Adjuvant Glycan Nanocarrier
- PMID: 35592309
- PMCID: PMC9110926
- DOI: 10.3389/fchem.2022.864206
A Toll-like Receptor-Activating, Self-Adjuvant Glycan Nanocarrier
Abstract
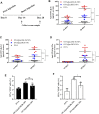
The global pandemic of COVID-19 highlights the importance of vaccination, which remains the most efficient measure against many diseases. Despite the progress in vaccine design, concerns with suboptimal antigen immunogenicity and delivery efficiency prevail. Self-adjuvant carriers-vehicles that can simultaneously deliver antigens and act as adjuvants-may improve efficacies in these aspects. Here, we developed a self-adjuvant carrier based on an acetyl glucomannan (acGM), which can activate toll-like receptor 2 (TLR2) and encapsulate the model antigen ovalbumin (OVA) via a double-emulsion process. In vitro tests showed that these OVA@acGM-8k nanoparticles (NPs) enhanced cellular uptake and activated TLR2 on the surface of dendritic cells (DCs), with increased expression of co-stimulatory molecules (e.g. CD80 and CD86) and pro-inflammatory cytokines (e.g. TNF-α and IL12p70). In vivo experiments in mice demonstrated that OVA@acGM-8k NPs accumulated in the lymph nodes and promoted DCs' maturation. The immunization also boosted the humoral and cellular immune responses. Our findings suggest that this self-adjuvant polysaccharide carrier could be a promising approach for vaccine development.
Keywords: glucomannan; polysaccharides; self-adjuvant; toll-like receptors; vaccination.
Copyright © 2022 Xie, Niu, Mu, Campos de Souza, Yin, Dong and Wang.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures







Similar articles
-
TLR2- and 4-independent immunomodulatory effect of high molecular weight components from Ascaris suum.Mol Immunol. 2014 Mar;58(1):17-26. doi: 10.1016/j.molimm.2013.10.011. Epub 2013 Nov 19. Mol Immunol. 2014. PMID: 24263181
-
Protein-bound polysaccharide activates dendritic cells and enhances OVA-specific T cell response as vaccine adjuvant.Immunobiology. 2013 Dec;218(12):1468-76. doi: 10.1016/j.imbio.2013.05.001. Epub 2013 May 13. Immunobiology. 2013. PMID: 23735481 Free PMC article.
-
Synergistic effect of dual targeting vaccine adjuvant with aminated β-glucan and CpG-oligodeoxynucleotides for both humoral and cellular immune responses.Acta Biomater. 2018 Sep 15;78:211-223. doi: 10.1016/j.actbio.2018.08.002. Epub 2018 Aug 9. Acta Biomater. 2018. PMID: 30098441
-
Angelica sinensis polysaccharide encapsulated into PLGA nanoparticles as a vaccine delivery and adjuvant system for ovalbumin to promote immune responses.Int J Pharm. 2019 Jan 10;554:72-80. doi: 10.1016/j.ijpharm.2018.11.008. Epub 2018 Nov 3. Int J Pharm. 2019. PMID: 30399435
-
Particulate formulations for the delivery of poly(I:C) as vaccine adjuvant.Adv Drug Deliv Rev. 2013 Oct;65(10):1386-99. doi: 10.1016/j.addr.2013.05.013. Epub 2013 Jun 7. Adv Drug Deliv Rev. 2013. PMID: 23751781 Review.
Cited by
-
A scaffold vaccine to promote tumor antigen cross-presentation via sustained toll-like receptor-2 (TLR2) activation.Bioact Mater. 2024 Apr 23;37:315-330. doi: 10.1016/j.bioactmat.2024.03.035. eCollection 2024 Jul. Bioact Mater. 2024. PMID: 38694764 Free PMC article.
-
Unraveling the pathogenic interplay between SARS-CoV-2 and polycystic ovary syndrome using bioinformatics and experimental validation.Sci Rep. 2024 Oct 2;14(1):22934. doi: 10.1038/s41598-024-74347-y. Sci Rep. 2024. PMID: 39358491 Free PMC article.
-
pH-responsive nano-vaccine combined with anti-PD-1 antibodies for enhanced immunotherapy of breast cancer.Theranostics. 2025 Apr 28;15(12):6022-6043. doi: 10.7150/thno.107200. eCollection 2025. Theranostics. 2025. PMID: 40365283 Free PMC article.
References
LinkOut - more resources
Full Text Sources

