Real-world study of multiple naloxone administration for opioid overdose reversal among bystanders
- PMID: 35596213
- PMCID: PMC9122081
- DOI: 10.1186/s12954-022-00627-3
Real-world study of multiple naloxone administration for opioid overdose reversal among bystanders
Abstract
Background: The increasing prevalence of highly potent, illicitly manufactured fentanyl and its analogues (IMF) in the USA is exacerbating the opioid epidemic which has worsened during the COVID-19 pandemic. Narcan® (naloxone HCl) Nasal Spray has been approved by the US Food and Drug Administration as a treatment for opioid-related overdoses. Due to the high potency of IMF, multiple naloxone administrations (MNA) may be needed per overdose event. It is essential to determine the patterns of naloxone use, including MNA, and preferences among bystanders who have used naloxone for opioid overdose reversal.
Methods: A cross-sectional web-based survey was administered to 125 adult US residents who administered 4 mg Narcan® Nasal Spray during an opioid overdose in the past year. The survey asked about the most recent overdose event, the use of Narcan® during the event and the associated withdrawal symptoms, and participant preferences regarding dosages of naloxone nasal spray. An open-ended voice survey was completed by 35 participants.
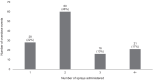
Results: Participants were mostly female (70%) and white (78%), while reported overdose events most frequently occurred in people who were males (54%) and white (86%). Most events (95%) were successfully reversed, with 78% using ≥ 2 doses and 30% using ≥ 3 doses of Narcan® Nasal Spray. Over 90% were worried that 1 Narcan® box may not be enough for a successful future reversal. Reported withdrawal symptoms were similar in overdose events where 1 versus ≥ 2 sprays were given. Eighty-six percent of participants reported more confidence in an 8 mg versus a 4 mg naloxone nasal spray and 77% reported a stronger preference for 8 mg over 4 mg.
Conclusions: MNA occurred in most overdose events, often involving more sprays than are provided in one Narcan® nasal spray box, and participants predominantly expressed having a stronger preference for and confidence in an 8 mg compared to a 4 mg nasal spray. This suggests the need and desire for a higher dose naloxone nasal spray formulation option. Given that bystanders may be the first to administer naloxone to someone experiencing an opioid overdose, ensuring access to an adequate naloxone supply is critical in addressing the opioid overdose epidemic.
Keywords: Bystander; COVID-19; Dosing; Fentanyl; Layperson; Multiple naloxone administrations; Naloxone; Narcan; Opioid overdose; Synthetic opioid.
© 2022. The Author(s).
Conflict of interest statement
Suzanne Carlberg-Racich has received consultancy fees from Hikma Community Health, a Subdivision of Hikma Specialty USA Inc. A. Raja Banerjee, Randa Abdelal and Neyla Darwaza have received stock options exercisable for, and other stock awards of, ordinary shares of Hikma Pharmaceuticals in the course of their employment at Hikma Pharmaceuticals. Diane Ito, Jessica Shoaff and Josh Epstein are employees of Stratevi, a consulting firm that has received research funding from Hikma Community Health, a Subdivision of Hikma Specialty USA Inc., to conduct this study.
Figures




References
-
- National Institute on Drug Abuse. Overdose death rates. 2021. https://www.drugabuse.gov/drug-topics/trends-statistics/overdose-death-r....
-
- Centers for Disease Control and Prevention. Overdose deaths and the involvement of illicit drugs. 2020. https://www.cdc.gov/drugoverdose/pubs/featured-topics/VS-overdose-deaths....
-
- Centers for Disease Control and Prevention. Vital Statistics Rapid Release; Provisional Drug Overdose Death Counts. 2021. https://www.cdc.gov/nchs/nvss/vsrr/drug-overdose-data.htm.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

