A data-driven model of brain volume changes in progressive supranuclear palsy
- PMID: 35602649
- PMCID: PMC9118104
- DOI: 10.1093/braincomms/fcac098
A data-driven model of brain volume changes in progressive supranuclear palsy
Abstract
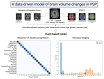
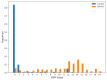
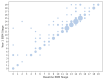
The most common clinical phenotype of progressive supranuclear palsy is Richardson syndrome, characterized by levodopa unresponsive symmetric parkinsonism, with a vertical supranuclear gaze palsy, early falls and cognitive impairment. There is currently no detailed understanding of the full sequence of disease pathophysiology in progressive supranuclear palsy. Determining the sequence of brain atrophy in progressive supranuclear palsy could provide important insights into the mechanisms of disease progression, as well as guide patient stratification and monitoring for clinical trials. We used a probabilistic event-based model applied to cross-sectional structural MRI scans in a large international cohort, to determine the sequence of brain atrophy in clinically diagnosed progressive supranuclear palsy Richardson syndrome. A total of 341 people with Richardson syndrome (of whom 255 had 12-month follow-up imaging) and 260 controls were included in the study. We used a combination of 12-month follow-up MRI scans, and a validated clinical rating score (progressive supranuclear palsy rating scale) to demonstrate the longitudinal consistency and utility of the event-based model's staging system. The event-based model estimated that the earliest atrophy occurs in the brainstem and subcortical regions followed by progression caudally into the superior cerebellar peduncle and deep cerebellar nuclei, and rostrally to the cortex. The sequence of cortical atrophy progresses in an anterior to posterior direction, beginning in the insula and then the frontal lobe before spreading to the temporal, parietal and finally the occipital lobe. This in vivo ordering accords with the post-mortem neuropathological staging of progressive supranuclear palsy and was robust under cross-validation. Using longitudinal information from 12-month follow-up scans, we demonstrate that subjects consistently move to later stages over this time interval, supporting the validity of the model. In addition, both clinical severity (progressive supranuclear palsy rating scale) and disease duration were significantly correlated with the predicted subject event-based model stage (P < 0.01). Our results provide new insights into the sequence of atrophy progression in progressive supranuclear palsy and offer potential utility to stratify people with this disease on entry into clinical trials based on disease stage, as well as track disease progression.
Keywords: biomarkers; disease progression; event-based model; machine learning; progressive supranuclear palsy.
© The Author(s) 2022. Published by Oxford University Press on behalf of the Guarantors of Brain.
Figures


References
-
- Schrag A, Ben-Shlomo Y, Quinn NP. Prevalence of progressive supranuclear palsy and multiple system atrophy: A cross-sectional study. Lancet. 1999;354(9192):1771–1775. - PubMed
-
- Steele JC, Richardson JC, Olszewski J. Progressive supranuclear palsy: A heterogeneous degeneration involving the brain stem, basal ganglia and cerebellum with vertical gaze and pseudobulbar palsy, nuchal dystonia and dementia. Arch Neurol. 1964;10(4):333–359. - PubMed
Grants and funding
- L30 AG069301/AG/NIA NIH HHS/United States
- MR/T046422/1/MRC_/Medical Research Council/United Kingdom
- U24 AG057437/AG/NIA NIH HHS/United States
- MR/T027770/1/MRC_/Medical Research Council/United Kingdom
- R01 AG038791/AG/NIA NIH HHS/United States
- MC_UU_00030/14/MRC_/Medical Research Council/United Kingdom
- U19 AG063911/AG/NIA NIH HHS/United States
- MR/M023664/1/MRC_/Medical Research Council/United Kingdom
- MR/T046015/1/MRC_/Medical Research Council/United Kingdom
- MR/S03546X/1/MRC_/Medical Research Council/United Kingdom
- MC_U105597119/MRC_/Medical Research Council/United Kingdom
- P01 AG019724/AG/NIA NIH HHS/United States
- 220258/WT_/Wellcome Trust/United Kingdom
- MR/P01271X/1/MRC_/Medical Research Council/United Kingdom
- MC_UU_00005/12/MRC_/Medical Research Council/United Kingdom
- MR/M008525/1/MRC_/Medical Research Council/United Kingdom
- K23 AG073514/AG/NIA NIH HHS/United States
