Bacterial Adaptation to Venom in Snakes and Arachnida
- PMID: 35604233
- PMCID: PMC9248900
- DOI: 10.1128/spectrum.02408-21
Bacterial Adaptation to Venom in Snakes and Arachnida
Abstract
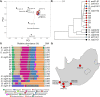
Animal venoms are considered sterile sources of antimicrobial compounds with strong membrane-disrupting activity against multidrug-resistant bacteria. However, venomous bite wound infections are common in developing nations. Investigating the envenomation organ and venom microbiota of five snake and two spider species, we observed venom community structures that depend on the host venomous animal species and evidenced recovery of viable microorganisms from black-necked spitting cobra (Naja nigricollis) and Indian ornamental tarantula (Poecilotheria regalis) venoms. Among the bacterial isolates recovered from N. nigricollis, we identified two venom-resistant, novel sequence types of Enterococcus faecalis whose genomes feature 16 virulence genes, indicating infectious potential, and 45 additional genes, nearly half of which improve bacterial membrane integrity. Our findings challenge the dogma of venom sterility and indicate an increased primary infection risk in the clinical management of venomous animal bite wounds. IMPORTANCE Notwithstanding their 3 to 5% mortality, the 2.7 million envenomation-related injuries occurring annually-predominantly across Africa, Asia, and Latin America-are also major causes of morbidity. Venom toxin-damaged tissue will develop infections in some 75% of envenomation victims, with E. faecalis being a common culprit of disease; however, such infections are generally considered to be independent of envenomation. Here, we provide evidence on venom microbiota across snakes and arachnida and report on the convergent evolution mechanisms that can facilitate adaptation to black-necked cobra venom in two independent E. faecalis strains, easily misidentified by biochemical diagnostics. Therefore, since inoculation with viable and virulence gene-harboring bacteria can occur during envenomation, acute infection risk management following envenomation is warranted, particularly for immunocompromised and malnourished victims in resource-limited settings. These results shed light on how bacteria evolve for survival in one of the most extreme environments on Earth and how venomous bites must be also treated for infections.
Keywords: drug resistance evolution; extremophiles; genome analysis; microbiome; multidrug resistance; venom.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Glaser HSR. 1948. Bactericidal activity of Crotalus venom in vitro. Copeia 1948:245. doi: 10.2307/1438710. - DOI
-
- Samy R, Gopalakrishnakone P, Satyanarayanajois S, Stiles B, Chow V. 2013. Snake venom proteins and peptides as novel antibiotics against microbial infections. Curr Proteomics 10:10–28. doi: 10.2174/1570164611310010003. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

