Multisensory task demands temporally extend the causal requirement for visual cortex in perception
- PMID: 35606448
- PMCID: PMC9126973
- DOI: 10.1038/s41467-022-30600-4
Multisensory task demands temporally extend the causal requirement for visual cortex in perception
Erratum in
-
Author Correction: Multisensory task demands temporally extend the causal requirement for visual cortex in perception.Nat Commun. 2022 Jul 14;13(1):4088. doi: 10.1038/s41467-022-31518-7. Nat Commun. 2022. PMID: 35835734 Free PMC article. No abstract available.
Abstract
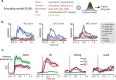
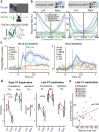
Primary sensory areas constitute crucial nodes during perceptual decision making. However, it remains unclear to what extent they mainly constitute a feedforward processing step, or rather are continuously involved in a recurrent network together with higher-order areas. We found that the temporal window in which primary visual cortex is required for the detection of identical visual stimuli was extended when task demands were increased via an additional sensory modality that had to be monitored. Late-onset optogenetic inactivation preserved bottom-up, early-onset responses which faithfully encoded stimulus features, and was effective in impairing detection only if it preceded a late, report-related phase of the cortical response. Increasing task demands were marked by longer reaction times and the effect of late optogenetic inactivation scaled with reaction time. Thus, independently of visual stimulus complexity, multisensory task demands determine the temporal requirement for ongoing sensory-related activity in V1, which overlaps with report-related activity.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures






Similar articles
-
Audiovisual Modulation in Mouse Primary Visual Cortex Depends on Cross-Modal Stimulus Configuration and Congruency.J Neurosci. 2017 Sep 6;37(36):8783-8796. doi: 10.1523/JNEUROSCI.0468-17.2017. Epub 2017 Aug 8. J Neurosci. 2017. PMID: 28821672 Free PMC article.
-
Perceptual Weighting of V1 Spikes Revealed by Optogenetic White Noise Stimulation.J Neurosci. 2022 Apr 13;42(15):3122-3132. doi: 10.1523/JNEUROSCI.1736-21.2022. Epub 2022 Mar 1. J Neurosci. 2022. PMID: 35232760 Free PMC article.
-
Occipital transcranial magnetic stimulation has opposing effects on visual and auditory stimulus detection: implications for multisensory interactions.J Neurosci. 2007 Oct 24;27(43):11465-72. doi: 10.1523/JNEUROSCI.2827-07.2007. J Neurosci. 2007. PMID: 17959789 Free PMC article.
-
Good times for multisensory integration: Effects of the precision of temporal synchrony as revealed by gamma-band oscillations.Neuropsychologia. 2007 Feb 1;45(3):561-71. doi: 10.1016/j.neuropsychologia.2006.01.013. Epub 2006 Mar 20. Neuropsychologia. 2007. PMID: 16542688
-
Playing the electric light orchestra--how electrical stimulation of visual cortex elucidates the neural basis of perception.Philos Trans R Soc Lond B Biol Sci. 2015 Sep 19;370(1677):20140206. doi: 10.1098/rstb.2014.0206. Philos Trans R Soc Lond B Biol Sci. 2015. PMID: 26240421 Free PMC article. Review.
Cited by
-
Testing the role of spontaneous activity in visuospatial perception with patterned optogenetics.PLoS One. 2025 Feb 27;20(2):e0318863. doi: 10.1371/journal.pone.0318863. eCollection 2025. PLoS One. 2025. PMID: 40014595 Free PMC article.
-
Protocol for synthesizing, implanting, and using polydimethylsiloxane as skull replacement in mice for imaging, electrophysiology, and optogenetics.STAR Protoc. 2025 Jul 22;6(3):103964. doi: 10.1016/j.xpro.2025.103964. Online ahead of print. STAR Protoc. 2025. PMID: 40700011 Free PMC article.
-
How 'visual' is the visual cortex? The interactions between the visual cortex and other sensory, motivational and motor systems as enabling factors for visual perception.Philos Trans R Soc Lond B Biol Sci. 2023 Sep 25;378(1886):20220336. doi: 10.1098/rstb.2022.0336. Epub 2023 Aug 7. Philos Trans R Soc Lond B Biol Sci. 2023. PMID: 37545313 Free PMC article. Review.
-
Human Brain Project Partnering Projects Meeting: Status Quo and Outlook.eNeuro. 2023 Sep 5;10(9):ENEURO.0091-23.2023. doi: 10.1523/ENEURO.0091-23.2023. Print 2023 Sep. eNeuro. 2023. PMID: 37669867 Free PMC article.
-
Adolescent-like Processing of Behaviorally Salient Cues in Sensory and Prefrontal Cortices of Adult Preterm-Born Mice.Res Sq [Preprint]. 2024 Dec 11:rs.3.rs-5529783. doi: 10.21203/rs.3.rs-5529783/v1. Res Sq. 2024. PMID: 39711564 Free PMC article. Preprint.
References
-
- Harris KD, Mrsic-Flogel TD. Cortical connectivity and sensory coding. Nature. 2013;503:51–58. - PubMed
-
- Crochet, S., Lee, S.-H. & Petersen, C. C. H. Neural circuits for goal-directed sensorimotor transformations. Trends Neurosci. 10.1016/j.tins.2018.08.011 (2018). - PubMed
-
- Supèr H, Spekreijse H, Lamme VAF. Two distinct modes of sensory processing observed in monkey primary visual cortex (V1) Nat. Neurosci. 2001;4:304–310. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

