The Cif proteins from Wolbachia prophage WO modify sperm genome integrity to establish cytoplasmic incompatibility
- PMID: 35609042
- PMCID: PMC9128985
- DOI: 10.1371/journal.pbio.3001584
The Cif proteins from Wolbachia prophage WO modify sperm genome integrity to establish cytoplasmic incompatibility
Update in
-
The mechanism of cytoplasmic incompatibility is conserved in Wolbachia-infected Aedes aegypti mosquitoes deployed for arbovirus control.PLoS Biol. 2024 Mar 28;22(3):e3002573. doi: 10.1371/journal.pbio.3002573. eCollection 2024 Mar. PLoS Biol. 2024. PMID: 38547237 Free PMC article.
Abstract
Inherited microorganisms can selfishly manipulate host reproduction to drive through populations. In Drosophila melanogaster, germline expression of the native Wolbachia prophage WO proteins CifA and CifB cause cytoplasmic incompatibility (CI) in which embryos from infected males and uninfected females suffer catastrophic mitotic defects and lethality; however, in infected females, CifA expression rescues the embryonic lethality and thus imparts a fitness advantage to the maternally transmitted Wolbachia. Despite widespread relevance to sex determination, evolution, and vector control, the mechanisms underlying when and how CI impairs male reproduction remain unknown and a topic of debate. Here, we use cytochemical, microscopic, and transgenic assays in D. melanogaster to demonstrate that CifA and CifB proteins of wMel localize to nuclear DNA throughout the process of spermatogenesis. Cif proteins cause abnormal histone retention in elongating spermatids and protamine deficiency in mature sperms that travel to the female reproductive tract with Cif proteins. Notably, protamine gene knockouts enhance wild-type CI. In ovaries, CifA localizes to germ cell nuclei and cytoplasm of early-stage egg chambers; however, Cifs are absent in late-stage oocytes and subsequently in fertilized embryos. Finally, CI and rescue are contingent upon a newly annotated CifA bipartite nuclear localization sequence. Together, our results strongly support the Host modification model of CI in which Cifs initially modify the paternal and maternal gametes to bestow CI-defining embryonic lethality and rescue.
Conflict of interest statement
I have read the journal’s policy and the authors of this manuscript have the following competing interests: SRB is listed as an inventor on a provisional patent related to this work.
Figures
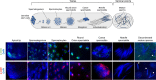
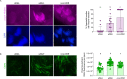

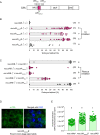
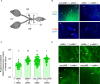
Comment in
-
Perplexing dynamics of Wolbachia proteins for cytoplasmic incompatibility.PLoS Biol. 2022 May 25;20(5):e3001644. doi: 10.1371/journal.pbio.3001644. eCollection 2022 May. PLoS Biol. 2022. PMID: 35613073 Free PMC article.
Similar articles
-
The mechanism of cytoplasmic incompatibility is conserved in Wolbachia-infected Aedes aegypti mosquitoes deployed for arbovirus control.PLoS Biol. 2024 Mar 28;22(3):e3002573. doi: 10.1371/journal.pbio.3002573. eCollection 2024 Mar. PLoS Biol. 2024. PMID: 38547237 Free PMC article.
-
Cytoplasmic incompatibility factor proteins from Wolbachia prophage are costly to sperm development in Drosophila melanogaster.Proc Biol Sci. 2025 Feb;292(2040):20243016. doi: 10.1098/rspb.2024.3016. Epub 2025 Feb 12. Proc Biol Sci. 2025. PMID: 39933580 Free PMC article.
-
The impacts of cytoplasmic incompatibility factor (cifA and cifB) genetic variation on phenotypes.Genetics. 2021 Mar 3;217(1):1-13. doi: 10.1093/genetics/iyaa007. Genetics. 2021. PMID: 33683351 Free PMC article.
-
The Biochemistry of Cytoplasmic Incompatibility Caused by Endosymbiotic Bacteria.Genes (Basel). 2020 Jul 25;11(8):852. doi: 10.3390/genes11080852. Genes (Basel). 2020. PMID: 32722516 Free PMC article. Review.
-
Symbiont-mediated cytoplasmic incompatibility: what have we learned in 50 years?Elife. 2020 Sep 25;9:e61989. doi: 10.7554/eLife.61989. Elife. 2020. PMID: 32975515 Free PMC article. Review.
Cited by
-
Wolbachia action in the sperm produces developmentally deferred chromosome segregation defects during the Drosophila mid-blastula transition.Elife. 2022 Sep 23;11:e81292. doi: 10.7554/eLife.81292. Elife. 2022. PMID: 36149408 Free PMC article.
-
The cellular lives of Wolbachia.Nat Rev Microbiol. 2023 Nov;21(11):750-766. doi: 10.1038/s41579-023-00918-x. Epub 2023 Jul 10. Nat Rev Microbiol. 2023. PMID: 37430172 Review.
-
Single-cell transcriptome sequencing reveals Wolbachia-mediated modification in early stages of Drosophila spermatogenesis.Proc Biol Sci. 2023 Jan 11;290(1990):20221963. doi: 10.1098/rspb.2022.1963. Epub 2023 Jan 11. Proc Biol Sci. 2023. PMID: 36629101 Free PMC article.
-
Intra-lineage microevolution of Wolbachia leads to the emergence of new cytoplasmic incompatibility patterns.PLoS Biol. 2024 Feb 5;22(2):e3002493. doi: 10.1371/journal.pbio.3002493. eCollection 2024 Feb. PLoS Biol. 2024. PMID: 38315724 Free PMC article.
-
The mechanism of cytoplasmic incompatibility is conserved in Wolbachia-infected Aedes aegypti mosquitoes deployed for arbovirus control.PLoS Biol. 2024 Mar 28;22(3):e3002573. doi: 10.1371/journal.pbio.3002573. eCollection 2024 Mar. PLoS Biol. 2024. PMID: 38547237 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

