Change in Urinary Myoinositol/Citrate Ratio Associates with Progressive Loss of Renal Function in ADPKD Patients
- PMID: 35613556
- PMCID: PMC9393825
- DOI: 10.1159/000524851
Change in Urinary Myoinositol/Citrate Ratio Associates with Progressive Loss of Renal Function in ADPKD Patients
Abstract
Introduction: In autosomal dominant polycystic kidney disease (ADPKD) patients, predicting renal disease progression is important to make a prognosis and to support the clinical decision whether to initiate renoprotective therapy. Conventional markers all have their limitations. Metabolic profiling is a promising strategy for risk stratification. We determined the prognostic performance to identify patients with a fast progressive disease course and evaluated time-dependent changes in urinary metabolites.
Methods: Targeted, quantitative metabolomics analysis (1H NMR-spectroscopy) was performed on spot urinary samples at two time points, baseline (n = 324, 61% female; mean age 45 years, SD 11; median eGFR 61 mL/min/1.73 m2, IQR 42-88; mean years of creatinine follow-up 3.7, SD 1.3) and a sample obtained after 3 years of follow-up (n = 112). Patients were stratified by their eGFR slope into fast and slow progressors based on an annualized change of > -3.0 or ≤ -3.0 mL/min/1.73 m2/year, respectively. Fifty-five urinary metabolites and ratios were quantified, and the significant ones were selected. Logistic regression was used to determine prognostic performance in identifying those with a fast progressive course using baseline urine samples. Repeated-measures ANOVA was used to analyze whether changes in urinary metabolites over a 3-year follow-up period differed between fast and slow progressors.
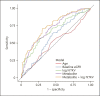
Results: In a single urinary sample, the prognostic performance of urinary metabolites was comparable to that of a model including height-adjusted total kidney volume (htTKV, AUC = 0.67). Combined with htTKV, the predictive value of the metabolite model increased (AUC = 0.75). Longitudinal analyses showed an increase in the myoinositol/citrate ratio (p < 0.001) in fast progressors, while no significant change was found in those with slow progression, which is in-line with an overall increase in the myoinositol/citrate ratio as GFR declines.
Conclusion: A metabolic profile, measured at a single time point, showed at least equivalent prognostic performance to an imaging-based risk marker in ADPKD. Changes in urinary metabolites over a 3-year follow-up period were associated with a fast progressive disease course.
Keywords: Autosomal dominant polycystic kidney disease; Biomarker; Estimated glomerular filtration rate slope; Progression; Urine metabolites.
© 2022 The Author(s). Published by S. Karger AG, Basel.
Conflict of interest statement
The authors have no conflicts of interest to declare.
Figures



References
-
- Neumann HP, Jilg C, Bacher J, Nabulsi Z, Malinoc A, Hummel B, et al. Epidemiology of autosomal-dominant polycystic kidney disease: an in-depth clinical study for south-western Germany. Nephrol Dial Transplant. 2013;28((6)):1472–87. - PubMed
-
- Torres VE, Harris PC, Pirson Y. Autosomal dominant polycystic kidney disease. Lancet. 2007;369((9569)):1287–301. - PubMed
-
- Spithoven EM, Kramer A, Meijer E, Orskov B, Wanner C, Abad JM, et al. Renal replacement therapy for autosomal dominant polycystic kidney disease (ADPKD) in Europe: prevalence and survival − an analysis of data from the ERA-EDTA Registry. Nephrol Dial Transplant. 2014;29((Suppl 4)):iv15–25. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

