Comparative Analysis of the Relative Fragmentation Stabilities of Polymorphic Alpha-Synuclein Amyloid Fibrils
- PMID: 35625557
- PMCID: PMC9138312
- DOI: 10.3390/biom12050630
Comparative Analysis of the Relative Fragmentation Stabilities of Polymorphic Alpha-Synuclein Amyloid Fibrils
Abstract
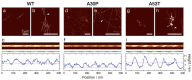
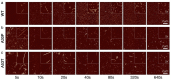
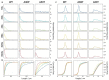
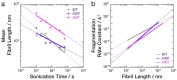
The division of amyloid fibril particles through fragmentation is implicated in the progression of human neurodegenerative disorders such as Parkinson's disease. Fragmentation of amyloid fibrils plays a crucial role in the propagation of the amyloid state encoded in their three-dimensional structures and may have an important role in the spreading of potentially pathological properties and phenotypes in amyloid-associated diseases. However, despite the mechanistic importance of fibril fragmentation, the relative stabilities of different types or different polymorphs of amyloid fibrils toward fragmentation remain to be quantified. We have previously developed an approach to compare the relative stabilities of different types of amyloid fibrils toward fragmentation. In this study, we show that controlled sonication, a widely used method of mechanical perturbation for amyloid seed generation, can be used as a form of mechanical perturbation for rapid comparative assessment of the relative fragmentation stabilities of different amyloid fibril structures. This approach is applied to assess the relative fragmentation stabilities of amyloid formed in vitro from wild type (WT) α-synuclein and two familial mutant variants of α-synuclein (A30P and A53T) that generate morphologically different fibril structures. Our results demonstrate that the fibril fragmentation stabilities of these different α-synuclein fibril polymorphs are all highly length dependent but distinct, with both A30P and A53T α-synuclein fibrils displaying increased resistance towards sonication-induced fibril fragmentation compared with WT α-synuclein fibrils. These conclusions show that fragmentation stabilities of different amyloid fibril polymorph structures can be diverse and suggest that the approach we report here will be useful in comparing the relative stabilities of amyloid fibril types or fibril polymorphs toward fragmentation under different biological conditions.
Keywords: amyloid; atomic force microscopy; fibril division; fibril fragmentation; image analysis; sonication; stability.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Structural features of α-synuclein amyloid fibrils revealed by Raman spectroscopy.J Biol Chem. 2018 Jan 19;293(3):767-776. doi: 10.1074/jbc.M117.812388. Epub 2017 Nov 30. J Biol Chem. 2018. PMID: 29191831 Free PMC article.
-
Dissociation of amyloid fibrils of alpha-synuclein and transthyretin by pressure reveals their reversible nature and the formation of water-excluded cavities.Proc Natl Acad Sci U S A. 2003 Aug 19;100(17):9831-6. doi: 10.1073/pnas.1734009100. Epub 2003 Aug 4. Proc Natl Acad Sci U S A. 2003. PMID: 12900507 Free PMC article.
-
α-Synuclein Fibril, Ribbon and Fibril-91 Amyloid Polymorphs Generation for Structural Studies.Methods Mol Biol. 2023;2551:345-355. doi: 10.1007/978-1-0716-2597-2_23. Methods Mol Biol. 2023. PMID: 36310214
-
Secondary Nucleation and the Conservation of Structural Characteristics of Amyloid Fibril Strains.Front Mol Biosci. 2021 Apr 16;8:669994. doi: 10.3389/fmolb.2021.669994. eCollection 2021. Front Mol Biosci. 2021. PMID: 33937341 Free PMC article. Review.
-
Molecular rules governing the structural polymorphism of amyloid fibrils in neurodegenerative diseases.Structure. 2023 Nov 2;31(11):1335-1347. doi: 10.1016/j.str.2023.08.006. Epub 2023 Aug 31. Structure. 2023. PMID: 37657437 Review.
Cited by
-
Mechanisms and pathology of protein misfolding and aggregation.Nat Rev Mol Cell Biol. 2023 Dec;24(12):912-933. doi: 10.1038/s41580-023-00647-2. Epub 2023 Sep 8. Nat Rev Mol Cell Biol. 2023. PMID: 37684425 Review.
-
The Amplification of Alpha-Synuclein Amyloid Fibrils is Suppressed under Fully Quiescent Conditions.Angew Chem Int Ed Engl. 2025 Feb 10;64(7):e202419173. doi: 10.1002/anie.202419173. Epub 2025 Jan 2. Angew Chem Int Ed Engl. 2025. PMID: 39648869 Free PMC article.
References
-
- Lovestam S., Schweighauser M., Matsubara T., Murayama S., Tomita T., Ando T., Hasegawa K., Yoshida M., Tarutani A., Hasegawa M., et al. Seeded assembly in vitro does not replicate the structures of alpha-synuclein filaments from multiple system atrophy. FEBS Open Biol. 2021;11:999–1013. doi: 10.1002/2211-5463.13110. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

