Incorporation of Oxidized Phenylalanine Derivatives into Insulin Signaling Relevant Proteins May Link Oxidative Stress to Signaling Conditions Underlying Chronic Insulin Resistance
- PMID: 35625712
- PMCID: PMC9138545
- DOI: 10.3390/biomedicines10050975
Incorporation of Oxidized Phenylalanine Derivatives into Insulin Signaling Relevant Proteins May Link Oxidative Stress to Signaling Conditions Underlying Chronic Insulin Resistance
Abstract
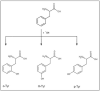
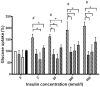
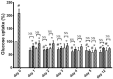
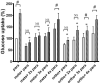
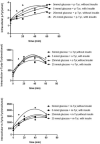
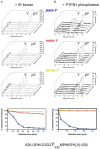
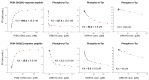
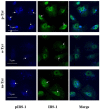
A link between oxidative stress and insulin resistance has been suggested. Hydroxyl free radicals are known to be able to convert phenylalanine (Phe) into the non-physiological tyrosine isoforms ortho- and meta-tyrosine (o-Tyr, m-Tyr). The aim of our study was to examine the role of o-Tyr and m-Tyr in the development of insulin resistance. We found that insulin-induced uptake of glucose was blunted in cultures of 3T3-L1 grown on media containing o- or m-Tyr. We show that these modified amino acids are incorporated into cellular proteins. We focused on insulin receptor substrate 1 (IRS-1), which plays a role in insulin signaling. The activating phosphorylation of IRS-1 was increased by insulin, the effect of which was abolished in cells grown in m-Tyr or o-Tyr media. We found that phosphorylation of m- or o-Tyr containing IRS-1 segments by insulin receptor (IR) kinase was greatly reduced, PTP-1B phosphatase was incapable of dephosphorylating phosphorylated m- or o-Tyr IRS-1 peptides, and the SH2 domains of phosphoinositide 3-kinase (PI3K) bound the o-Tyr IRS-1 peptides with greatly reduced affinity. According to our data, m- or o-Tyr incorporation into IRS-1 modifies its protein-protein interactions with regulating enzymes and effectors, thus IRS-1 eventually loses its capacity to play its role in insulin signaling, leading to insulin resistance.
Keywords: IRS-1; dephosphorylation; hydroxyl free radical; insulin resistance; meta-tyrosine; ortho-tyrosine; oxidative stress; phosphorylation.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures





Similar articles
-
Tyrosine isomers and hormonal signaling: A possible role for the hydroxyl free radical in insulin resistance.World J Diabetes. 2015 Apr 15;6(3):500-7. doi: 10.4239/wjd.v6.i3.500. World J Diabetes. 2015. PMID: 25897359 Free PMC article. Review.
-
Insulin-like growth factor-1 (IGF-1) receptor-insulin receptor substrate complexes in the uterus. Altered signaling response to estradiol in the IGF-1(m/m) mouse.J Biol Chem. 1998 May 8;273(19):11962-9. doi: 10.1074/jbc.273.19.11962. J Biol Chem. 1998. PMID: 9565625
-
Dephosphorylation of insulin receptor substrate 1 by the tyrosine phosphatase PTP2C.J Biol Chem. 1994 Jun 3;269(22):15833-7. J Biol Chem. 1994. PMID: 7515062
-
Reciprocal feedback regulation of insulin receptor and insulin receptor substrate tyrosine phosphorylation by phosphoinositide 3-kinase in primary adipocytes.Biochem J. 2002 Dec 15;368(Pt 3):875-84. doi: 10.1042/BJ20020903. Biochem J. 2002. PMID: 12220227 Free PMC article.
-
The new elements of insulin signaling. Insulin receptor substrate-1 and proteins with SH2 domains.Diabetes. 1993 May;42(5):643-50. doi: 10.2337/diab.42.5.643. Diabetes. 1993. PMID: 8387037 Review.
Cited by
-
Incident Cancer Risk in Patients with Incident Type 2 Diabetes Mellitus in Hungary (Part 1).Cancers (Basel). 2024 Apr 29;16(9):1745. doi: 10.3390/cancers16091745. Cancers (Basel). 2024. PMID: 38730697 Free PMC article.
-
The association between circulating phenylalanine and the temporal risk of impaired insulin markers in gestational diabetes mellitus.Mol Genet Metab Rep. 2024 Jun 14;40:101090. doi: 10.1016/j.ymgmr.2024.101090. eCollection 2024 Sep. Mol Genet Metab Rep. 2024. PMID: 38974841 Free PMC article.
-
Diabetes: A Multifaceted Disorder.Biomedicines. 2022 Jul 14;10(7):1698. doi: 10.3390/biomedicines10071698. Biomedicines. 2022. PMID: 35885003 Free PMC article.
-
The Impact of SGLT-2 Inhibitors on Hydroxyl Radical Markers and Diabetic Neuropathy: A Short-Term Clinical Study.Antioxidants (Basel). 2025 Feb 28;14(3):289. doi: 10.3390/antiox14030289. Antioxidants (Basel). 2025. PMID: 40227261 Free PMC article.
-
The relationship between hypoxia and Alzheimer's disease: an updated review.Front Aging Neurosci. 2024 Jul 17;16:1402774. doi: 10.3389/fnagi.2024.1402774. eCollection 2024. Front Aging Neurosci. 2024. PMID: 39086755 Free PMC article. Review.
References
-
- Galiero R., Caturano A., Vetrano E., Cesaro A., Rinaldi L., Salvatore T., Marfella R., Sardu C., Moscarella E., Gragnano F., et al. Pathophysiological mechanisms and clinical evidence of relationship between Nonalcoholic fatty liver disease (NAFLD) and cardiovascular disease. Rev. Cardiovasc. Med. 2021;22:755–768. doi: 10.31083/j.rcm2203082. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

