Impact of HER2 Status on Pathological Response after Neoadjuvant Chemotherapy in Early Triple-Negative Breast Cancer
- PMID: 35626113
- PMCID: PMC9139240
- DOI: 10.3390/cancers14102509
Impact of HER2 Status on Pathological Response after Neoadjuvant Chemotherapy in Early Triple-Negative Breast Cancer
Abstract
Purpose: Investigates the link between HER2 status and histological response after neoadjuvant chemotherapy in patients with early TNBC.
Methods: We retrieved clinical and anatomopathological data retrospectively from 449 patients treated for the first time with standard neoadjuvant chemotherapy for early unilateral BC between 2005 and 2020. The primary endpoint was pathological complete response (pCR, i.e., ypT0 ypN0), according to HER2 status. Secondary endpoints included invasive disease-free survival (I-DFS) and overall survival (OS).
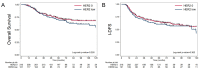
Results: 437 patients were included, and 121 (27.7%) patients had HER2-low tumours. The pCR rate was not significantly different between the HER2-low group vs. the HER2-0 group (35.7% versus 41.8%, p = 0.284) in either univariate analysis or multivariate analysis adjusted for TNM classification and grade (odds ratio [OR] = 0.70, confidence interval [CI] 95% 0.45-1.08). With a median follow-up of 72.9 months, no significant survival differences were observed between patients with HER2-low tumours vs. patients with HER2-0 tumours in terms of I-DFS (p = 0.487) and OS (p = 0.329).
Conclusions: In our cohort, HER2 status was not significantly associated with pCR in a manner consistent with data published recently on TNBC. However, the prognostic impact of HER2-low expression among TNBC patients warrants further evaluation.
Keywords: HER2-low; complete histologic response; prognosis; triple-negative breast cancer.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Freres P., Collignon J., Gennigens C., Scagnol I., Rorive A., Barbeaux A., Coucke P.A. Jérusalem Gle cancer du sein «triple né-gatif». Rev. Med. Liège. 2010;65:120–126. - PubMed
-
- Nabholtz J.M., Abrial C., Mouret-Reynier M.-A., Dauplat M.M., Weber B., Gligorov J., Forest A.M., Tredan O., Vanlemmens L., Petit T., et al. Multicentric neoadjuvant phase II study of panitumumab combined with an anthracycline/taxane-based chemotherapy in operable triple-negative breast cancer: Identification of biologically defined signatures predicting treatment impact. Ann. Oncol. 2014;25:1570–1577. doi: 10.1093/annonc/mdu183. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous