Interaction of Arginine-Rich Cell-Penetrating Peptides with an Artificial Neuronal Membrane
- PMID: 35626677
- PMCID: PMC9139471
- DOI: 10.3390/cells11101638
Interaction of Arginine-Rich Cell-Penetrating Peptides with an Artificial Neuronal Membrane
Abstract
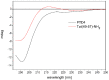
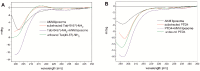
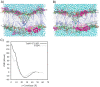
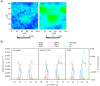
Arginine-rich cell-penetrating peptides (RRCPPs) exhibit intrinsic neuroprotective effects on neurons injured by acute ischemic stroke. Conformational properties, interaction, and the ability to penetrate the neural membrane are critical for the neuroprotective effects of RRCCPs. In this study, we applied circular dichroism (CD) spectroscopy and coarse-grained molecular dynamics (CG MD) simulations to investigate the interactions of two RRCPPs, Tat(49-57)-NH2 (arginine-rich motif of Tat HIV-1 protein) and PTD4 (a less basic Ala-scan analog of the Tat peptide), with an artificial neuronal membrane (ANM). CD spectra showed that in an aqueous environment, such as phosphate-buffered saline, the peptides mostly adopted a random coil (PTD4) or a polyproline type II helical (Tat(49-57)-NH2) conformation. On the other hand, in the hydrophobic environment of the ANM liposomes, the peptides showed moderate conformational changes, especially around 200 nm, as indicated by CD curves. The changes induced by the liposomes were slightly more significant in the PTD4 peptide. However, the nature of the conformational changes could not be clearly defined. CG MD simulations showed that the peptides are quickly attracted to the neuronal lipid bilayer and bind preferentially to monosialotetrahexosylganglioside (DPG1) molecules. However, the peptides did not penetrate the membrane even at increasing concentrations. This suggests that the energy barrier required to break the strong peptide-lipid electrostatic interactions was not exceeded in the simulated models. The obtained results show a correlation between the potential of mean force parameter and a peptide's cell membrane-penetrating ability and neuroprotective properties.
Keywords: PTD4 peptide; Tat(49–57)-NH2; artificial neuronal membrane; cell-penetrating peptides; circular dichroism studies; liposome; molecular dynamics simulations.
Conflict of interest statement
The authors declare that they have no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures

Similar articles
-
PTD4 Peptide Increases Neural Viability in an In Vitro Model of Acute Ischemic Stroke.Int J Mol Sci. 2021 Jun 4;22(11):6086. doi: 10.3390/ijms22116086. Int J Mol Sci. 2021. PMID: 34200045 Free PMC article.
-
Neuroprotective peptides fused to arginine-rich cell penetrating peptides: Neuroprotective mechanism likely mediated by peptide endocytic properties.Pharmacol Ther. 2015 Sep;153:36-54. doi: 10.1016/j.pharmthera.2015.06.002. Epub 2015 Jun 3. Pharmacol Ther. 2015. PMID: 26048328 Review.
-
Photodamage of lipid bilayers by irradiation of a fluorescently labeled cell-penetrating peptide.Biochim Biophys Acta. 2014 Jan;1840(1):507-15. doi: 10.1016/j.bbagen.2013.10.011. Epub 2013 Oct 14. Biochim Biophys Acta. 2014. PMID: 24135456 Free PMC article.
-
The neuroprotective efficacy of cell-penetrating peptides TAT, penetratin, Arg-9, and Pep-1 in glutamic acid, kainic acid, and in vitro ischemia injury models using primary cortical neuronal cultures.Cell Mol Neurobiol. 2014 Mar;34(2):173-81. doi: 10.1007/s10571-013-9999-3. Epub 2013 Nov 9. Cell Mol Neurobiol. 2014. PMID: 24213248 Free PMC article.
-
Antimicrobial and cell-penetrating peptides: structure, assembly and mechanisms of membrane lysis via atomistic and coarse-grained molecular dynamics simulations.Protein Pept Lett. 2010 Nov;17(11):1313-27. doi: 10.2174/0929866511009011313. Protein Pept Lett. 2010. PMID: 20673230 Review.
Cited by
-
New Conjugates of Vancomycin with Cell-Penetrating Peptides-Synthesis, Antimicrobial Activity, Cytotoxicity, and BBB Permeability Studies.Molecules. 2024 Nov 22;29(23):5519. doi: 10.3390/molecules29235519. Molecules. 2024. PMID: 39683678 Free PMC article.
-
Exploring the Chemical Features and Biomedical Relevance of Cell-Penetrating Peptides.Int J Mol Sci. 2024 Dec 25;26(1):59. doi: 10.3390/ijms26010059. Int J Mol Sci. 2024. PMID: 39795918 Free PMC article. Review.
-
Approaches for evaluation of novel CPP-based cargo delivery systems.Front Pharmacol. 2022 Oct 21;13:1056467. doi: 10.3389/fphar.2022.1056467. eCollection 2022. Front Pharmacol. 2022. PMID: 36339538 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

