Sepsis Encephalopathy Is Partly Mediated by miR370-3p-Induced Mitochondrial Injury but Attenuated by BAM15 in Cecal Ligation and Puncture Sepsis Male Mice
- PMID: 35628259
- PMCID: PMC9141734
- DOI: 10.3390/ijms23105445
Sepsis Encephalopathy Is Partly Mediated by miR370-3p-Induced Mitochondrial Injury but Attenuated by BAM15 in Cecal Ligation and Puncture Sepsis Male Mice
Abstract
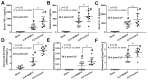
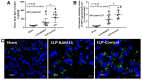
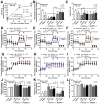
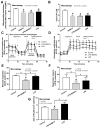
BAM15 (a mitochondrial uncoupling agent) was tested on cecal ligation and puncture (CLP) sepsis mice with in vitro experiments. BAM15 attenuated sepsis as indicated by survival, organ histology (kidneys and livers), spleen apoptosis (activated caspase 3), brain injury (SHIRPA score, serum s100β, serum miR370-3p, brain miR370-3p, brain TNF-α, and apoptosis), systemic inflammation (cytokines, cell-free DNA, endotoxemia, and bacteremia), and blood-brain barrier (BBB) damage (Evan's blue dye and the presence of green fluorescent E. coli in brain after an oral administration). In parallel, brain miR arrays demonstrated miR370-3p at 24 h but not 120 h post-CLP, which was correlated with metabolic pathways. Either lipopolysaccharide (LPS) or TNF-α upregulated miR370-3p in PC12 (neuron cells). An activation by sepsis factors (LPS, TNF-α, or miR370-3p transfection) damaged mitochondria (fluorescent color staining) and reduced cell ATP, possibly through profound mitochondrial activity (extracellular flux analysis) that was attenuated by BAM15. In bone-marrow-derived macrophages, LPS caused mitochondrial injury, decreased cell ATP, enhanced glycolysis activity (extracellular flux analysis), and induced pro-inflammatory macrophages (iNOS and IL-1β) which were neutralized by BAM15. In conclusion, BAM15 attenuated sepsis through decreased mitochondrial damage, reduced neuronal miR370-3p upregulation, and induced anti-inflammatory macrophages. BAM15 is proposed to be used as an adjuvant therapy against sepsis hyperinflammation.
Keywords: BAM15; cecal ligation and puncture; extracellular flux; sepsis; uncoupling agent.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

