Anti-SARS-CoV-2 Titers Predict the Severity of COVID-19
- PMID: 35632830
- PMCID: PMC9143418
- DOI: 10.3390/v14051089
Anti-SARS-CoV-2 Titers Predict the Severity of COVID-19
Abstract
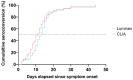
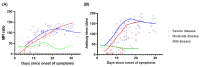
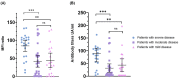
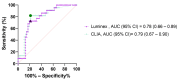
Coronavirus disease 2019 (COVID-19) due to SARS-CoV-2 is associated with a wide spectrum of disease, ranging from asymptomatic infection to acute respiratory distress syndrome. Some biomarkers may predict disease severity. Among them, the anti-SARS-CoV-2 antibody response has been related to severe disease. The aim of this study was to assess the correlation between the anti-SARS-CoV-2 serological response and COVID-19 outcome. Demographic, clinical, and biological data from nasopharyngeal-PCR confirmed COVID-19 hospitalized patients were prospectively collected between April and August 2020 at our institution. All patients had serial weekly serology testing for a maximum of three blood samples or until discharge. Two different serological assays were used: a chemiluminescent assay and an in-house developed Luminex immunoassay. Kinetics of the serological response and correlation between the antibody titers and outcome were assessed. Among the 70 patients enrolled in the study, 22 required invasive ventilation, 29 required non-invasive ventilation or oxygen supplementation, and 19 did not require any oxygen supplementation. Median duration of symptoms upon admission for the three groups were 13, 8, and 9 days, respectively. Antibody titers gradually increased for up to 3 weeks since the onset of symptoms for patients requiring oxygen supplementation with significantly higher antibody titers for patients requiring invasive ventilation. Antibody titers on admission were also significantly higher in severely ill patients and serology performed well in predicting the necessity of invasive ventilation (AUC: 0.79, 95% CI: 0.67-0.9). Serology testing at admission may be a good indicator to identify severe COVID-19 patients who will require invasive mechanical ventilation.
Keywords: COVID-19; SARS-CoV-2; antibody response; disease severity; outcome; serology.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Richardson S., Hirsch J.S., Narasimhan M., Crawford J.M., McGinn T., Davidson K.W., The Northwell COVID-19 Research Consortium Presenting Characteristics, Comorbidities, and Outcomes Among 5700 Patients Hospitalized with COVID-19 in the New York City Area. JAMA. 2020;323:2052–2059. doi: 10.1001/jama.2020.6775. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

