Normative intracranial EEG maps epileptogenic tissues in focal epilepsy
- PMID: 35640886
- PMCID: PMC9630716
- DOI: 10.1093/brain/awab480
Normative intracranial EEG maps epileptogenic tissues in focal epilepsy
Erratum in
-
Correction to: Normative intracranial EEG maps epileptogenic tissues in focal epilepsy.Brain. 2023 Jan 5;146(1):e5. doi: 10.1093/brain/awac337. Brain. 2023. PMID: 36240325 Free PMC article. No abstract available.
Abstract
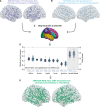
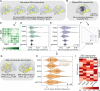
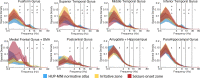
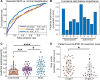
Planning surgery for patients with medically refractory epilepsy often requires recording seizures using intracranial EEG. Quantitative measures derived from interictal intracranial EEG yield potentially appealing biomarkers to guide these surgical procedures; however, their utility is limited by the sparsity of electrode implantation as well as the normal confounds of spatiotemporally varying neural activity and connectivity. We propose that comparing intracranial EEG recordings to a normative atlas of intracranial EEG activity and connectivity can reliably map abnormal regions, identify targets for invasive treatment and increase our understanding of human epilepsy. Merging data from the Penn Epilepsy Center and a public database from the Montreal Neurological Institute, we aggregated interictal intracranial EEG retrospectively across 166 subjects comprising >5000 channels. For each channel, we calculated the normalized spectral power and coherence in each canonical frequency band. We constructed an intracranial EEG atlas by mapping the distribution of each feature across the brain and tested the atlas against data from novel patients by generating a z-score for each channel. We demonstrate that for seizure onset zones within the mesial temporal lobe, measures of connectivity abnormality provide greater distinguishing value than univariate measures of abnormal neural activity. We also find that patients with a longer diagnosis of epilepsy have greater abnormalities in connectivity. By integrating measures of both single-channel activity and inter-regional functional connectivity, we find a better accuracy in predicting the seizure onset zones versus normal brain (area under the curve = 0.77) compared with either group of features alone. We propose that aggregating normative intracranial EEG data across epilepsy centres into a normative atlas provides a rigorous, quantitative method to map epileptic networks and guide invasive therapy. We publicly share our data, infrastructure and methods, and propose an international framework for leveraging big data in surgical planning for refractory epilepsy.
Keywords: brain network model; epilepsy; epilepsy surgery; functional connectivity; intracranial EEG.
© The Author(s) 2022. Published by Oxford University Press on behalf of the Guarantors of Brain. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Figures



References
-
- Kwan P, Schachter SC, Brodie MJ. Drug-resistant epilepsy. N Engl J Med. 2011;365:919–926. - PubMed
-
- Téllez-Zenteno JF, Dhar R, Wiebe S. Long-term seizure outcomes following epilepsy surgery: a systematic review and meta-analysis. Brain. 2005;128(5):1188–1198. - PubMed
-
- Wiebe S, Blume WT, Girvin JP, Eliasziw M. A randomized, controlled trial of surgery for temporal-lobe epilepsy. N Engl J Med. 2001;345(5):311–318. - PubMed

