Extension of cervical screening intervals with primary human papillomavirus testing: observational study of English screening pilot data
- PMID: 35640960
- PMCID: PMC9153243
- DOI: 10.1136/bmj-2021-068776
Extension of cervical screening intervals with primary human papillomavirus testing: observational study of English screening pilot data
Abstract
Objectives: To provide updated evidence about the risk of cervical intraepithelial neoplasia grade 3 or higher (CIN3+) and cervical cancer after a negative human papillomavirus (HPV) test in primary cervical screening, by age group and test assay.
Design: Observational study.
Setting: Real world data from the English HPV screening pilot's first and second rounds (2013-16, follow-up to end of 2019).
Participants: 1 341 584 women.
Interventions: Cervical screening with HPV testing or liquid based cytological testing (cytology or smear tests). Women screened with cytology were referred to colposcopy after high grade cytological abnormalities or after borderline or low grade abnormalities combined with a positive HPV triage test. Women screened with HPV testing who were positive were referred at baseline if their cytology triage test showed at least borderline abnormalities or after a retest (early recall) at 12 and 24 months if they had persistent abnormalities.
Main outcome measures: Detection of CIN3+ and cervical cancer after a negative HPV test.
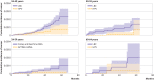
Results: For women younger than 50 years, second round detection of CIN3+ in this study was significantly lower after a negative HPV screen in the first round than after cytology testing (1.21/1000 v 4.52/1000 women screened, adjusted odds ratio 0.26, 95% confidence interval 0.23 to 0.30), as was the risk of interval cervical cancer (1.31/100 000 v 2.90/100 000 woman years, adjusted hazard ratio 0.44, 0.23 to 0.84). Risk of an incident CIN3+ detected at the second screening round in the pilot five years after a negative HPV test was even lower in women older than 50 years, than in three years in women younger than 50 years (0.57/1000 v 1.21/1000 women screened, adjusted odds ratio 0.46, 0.27 to 0.79). Women with negative HPV tests at early recall after a positive HPV screening test without cytological abnormalities had a higher detection rate of CIN3+ at the second routine recall than women who initially tested HPV negative (5.39/1000 v 1.21/1000 women screened, adjusted odds ratio 3.27, 95% confidence interval 2.21 to 4.84). Detection after a negative result on a clinically validated APTIMA mRNA HPV test was similar to that after clinically validated cobas and RealTime DNA tests (for CIN3+ at the second round 1.32/1000 v 1.14/1000 women screened, adjusted odds ratio 1.05, 0.73 to 1.50).
Conclusions: These data support an extension of the screening intervals, regardless of the test assay used: to five years after a negative HPV test in women aged 25-49 years, and even longer for women aged 50 years and older. The screening interval for HPV positive women who have negative HPV tests at early recall should be kept at three years.
© Author(s) (or their employer(s)) 2019. Re-use permitted under CC BY. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: All authors have completed the ICMJE uniform disclosure form at ww.icmje.org/coi_disclosure.pdf and declare: MR was provided Public Health England funding for the epidemiological evaluation of the pilot, and an unrelated study; is a member of the Public Health England Laboratory Technology Group, HPV Development Group, and HPV Self-sampling Operational Steering Group and Project Board; attended meetings with various HPV assay manufacturers; declares fee for lecture from Hologic, paid to employer. KC declares research funding or consumables to support research from Cepheid, Euroimmun, GeneFirst, SelfScreen, Hiantis Seegene, Roche, Abbott, and Hologic, paid to employer; is a professional clinical advisor to Public Health England; is a member of Public Health England Laboratory Technology Group and HPV Self-sampling Operational Steering Group. CM held an honorary appointment at Public Health England to process the data for the pilot. FP declares no competing interests. KD is an adviser to Public Health England (this position is funded by Public Health England as a secondment from her main employment); chairs the Public Health England Laboratory Clinical Professional Group, the HPV Development Group, and several groups related to the evaluation of self-sampling; was a consultant to the Scally Review of cervical screening in Ireland and the RCOG review of cervical cancer audit in Ireland, both completed in 2019; has provided expert medicolegal reports prepared for claimants and defendants including in cases of cervical cancer; received support with travel expenses to attend an international meeting in May 2019 from Hologic, a company manufacturing equipment and consumables for cytology and HPV testing. HK is former chair of the Public Health England Advisory Committee for Cervical Screening. We report no other relationships or activities that could appear to have influenced the submitted work. The views expressed in this manuscript are those of the authors and do not represent the view of Public Health England. Patient consent: Women participating in the HPV primary screening pilot were invited to make an informed choice on participating in the cervical screening programme. A decision is made to accept or decline a screening test based on access to accurate and up-to-date information on the condition being screened for, the testing process and potential outcomes. Specific information was provided at the invitation stage allowing for personalised informed choice. Further opportunity was given to reflect on what the test and its results might mean when women attended for screening with the clinician taking the sample. Regulation 5, Health Service Regulations 2002, Confidentiality Advisory Group Reference 15/CAG/0207, was the legal basis to process the data.
Figures
Comment in
-
HPV screening for cervical cancer is reaching maturity.BMJ. 2022 May 31;377:o1303. doi: 10.1136/bmj.o1303. BMJ. 2022. PMID: 35640961 No abstract available.
References
-
- UK National Screening Committee. Cervical Screening Programme modifications looking at; interval/ surveillance, women over 64 and self-sampling (27 February 2019). https://legacyscreening.phe.org.uk/policydb_download.php?doc=1188.
