The relayed nuclear Overhauser effect in magnetization transfer and chemical exchange saturation transfer MRI
- PMID: 35642102
- PMCID: PMC9708952
- DOI: 10.1002/nbm.4778
The relayed nuclear Overhauser effect in magnetization transfer and chemical exchange saturation transfer MRI
Abstract
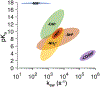
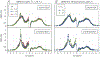
Magnetic resonance (MR) is a powerful technique for noninvasively probing molecular species in vivo but suffers from low signal sensitivity. Saturation transfer (ST) MRI approaches, including chemical exchange saturation transfer (CEST) and conventional magnetization transfer contrast (MTC), allow imaging of low-concentration molecular components with enhanced sensitivity using indirect detection via the abundant water proton pool. Several recent studies have shown the utility of chemical exchange relayed nuclear Overhauser effect (rNOE) contrast originating from nonexchangeable carbon-bound protons in mobile macromolecules in solution. In this review, we describe the mechanisms leading to the occurrence of rNOE-based signals in the water saturation spectrum (Z-spectrum), including those from mobile and immobile molecular sources and from molecular binding. While it is becoming clear that MTC is mainly an rNOE-based signal, we continue to use the classical MTC nomenclature to separate it from the rNOE signals of mobile macromolecules, which we will refer to as rNOEs. Some emerging applications of the use of rNOEs for probing macromolecular solution components such as proteins and carbohydrates in vivo or studying the binding of small substrates are discussed.
Keywords: Z-spectrum; chemical exchange saturation transfer; dipolar cross relaxation; magnetization transfer contrast; molecular binding; molecular imaging; nuclear Overhauser effect; signal enhancement.
© 2022 The Authors. NMR in Biomedicine published by John Wiley & Sons Ltd.
Figures



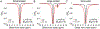



References
-
- Wolff SD, Balaban RS. NMR imaging of labile proton exchange. J Magn Reson. 1990;86(1):164–169.
-
- Wolff SD, Balaban RS. Magnetization transfer contrast (MTC) and tissue water proton relaxation in vivo. Magn Reson Med. 1989;10(1):135–144. - PubMed
-
- Ward K, Aletras A, Balaban RS. A new class of contrast agents for MRI based on proton chemical exchange dependent saturation transfer (CEST). J Magn Reson. 2000;143(1):79–87. - PubMed
-
- van Zijl PCM, Sehgal AA. Proton Chemical Exchange Saturation Transfer (CEST) MRS and MRI. In: eMagRes.2016:1307–1332.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

