Lymphocyte crosstalk is required for monocyte-intrinsic trained immunity to Plasmodium falciparum
- PMID: 35642634
- PMCID: PMC9151696
- DOI: 10.1172/JCI139298
Lymphocyte crosstalk is required for monocyte-intrinsic trained immunity to Plasmodium falciparum
Abstract
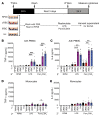
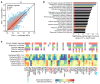
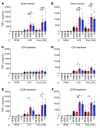
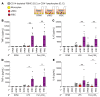
Plasmodium falciparum (P. falciparum) induces trained innate immune responses in vitro, where initial stimulation of adherent PBMCs with P. falciparum-infected RBCs (iRBCs) results in hyperresponsiveness to subsequent ligation of TLR2. This response correlates with the presence of T and B lymphocytes in adherent PBMCs, suggesting that innate immune training is partially due to adaptive immunity. We found that T cell-depleted PBMCs and purified monocytes alone did not elicit hyperproduction of IL-6 and TNF-α under training conditions. Analysis of P. falciparum-trained PBMCs showed that DCs did not develop under control conditions, and IL-6 and TNF-α were primarily produced by monocytes and DCs. Transwell experiments isolating purified monocytes from either PBMCs or purified CD4+ T cells, but allowing diffusion of secreted proteins, enabled monocytes trained with iRBCs to hyperproduce IL-6 and TNF-α after TLR restimulation. Purified monocytes stimulated with IFN-γ hyperproduced IL-6 and TNF-α, whereas blockade of IFN-γ in P. falciparum-trained PBMCs inhibited trained responses. Assay for transposase-accessible chromatin with high-throughput sequencing (ATAC-Seq) on monocytes from patients with malaria showed persistently open chromatin at genes that appeared to be trained in vitro. Together, these findings indicate that the trained immune response of monocytes to P. falciparum is not completely cell intrinsic but depends on soluble signals from lymphocytes.
Keywords: Immunology; Infectious disease; Innate immunity; Malaria; T cells.
Figures



Similar articles
-
IL-12 producing monocytes and IFN-gamma and TNF-alpha producing T-lymphocytes are increased in placentas infected by Plasmodium falciparum.J Reprod Immunol. 2007 Jun;74(1-2):152-62. doi: 10.1016/j.jri.2006.10.001. Epub 2006 Dec 27. J Reprod Immunol. 2007. PMID: 17194481
-
Influence of tumor necrosis factor-alpha on the ability of monocytes and lymphocytes to destroy intraerythrocytic Plasmodium falciparum in vitro.Cell Immunol. 2001 Mar 15;208(2):73-9. doi: 10.1006/cimm.2001.1770. Cell Immunol. 2001. PMID: 11333139
-
Plasmodium falciparum-free merozoites and infected RBCs distinctly affect soluble CD40 ligand-mediated maturation of immature monocyte-derived dendritic cells.J Leukoc Biol. 2008 Jul;84(1):244-54. doi: 10.1189/jlb.0807565. Epub 2008 Apr 15. J Leukoc Biol. 2008. PMID: 18413701
-
Selected problems of malaria blood stage immunity.Tokai J Exp Clin Med. 1998 Apr;23(2):55-62. Tokai J Exp Clin Med. 1998. PMID: 10021776 Review.
-
Human Vγ9Vδ2 T Lymphocytes in the Immune Response to P. falciparum Infection.Front Immunol. 2018 Nov 27;9:2760. doi: 10.3389/fimmu.2018.02760. eCollection 2018. Front Immunol. 2018. PMID: 30538708 Free PMC article. Review.
Cited by
-
Potent induction of trained immunity by Saccharomyces cerevisiae β-glucans.Front Immunol. 2024 Feb 13;15:1323333. doi: 10.3389/fimmu.2024.1323333. eCollection 2024. Front Immunol. 2024. PMID: 38415247 Free PMC article.
-
Trained immunity is regulated by T cell-induced CD40-TRAF6 signaling.Cell Rep. 2024 Sep 24;43(9):114664. doi: 10.1016/j.celrep.2024.114664. Epub 2024 Aug 22. Cell Rep. 2024. PMID: 39178113 Free PMC article.
-
Clinical immunity to malaria involves epigenetic reprogramming of innate immune cells.PNAS Nexus. 2024 Aug 6;3(8):pgae325. doi: 10.1093/pnasnexus/pgae325. eCollection 2024 Aug. PNAS Nexus. 2024. PMID: 39161730 Free PMC article.
-
Heat-killed Mycobacterium tuberculosis induces trained immunity in vitro and in vivo administered systemically or intranasally.iScience. 2024 Jan 11;27(2):108869. doi: 10.1016/j.isci.2024.108869. eCollection 2024 Feb 16. iScience. 2024. PMID: 38318361 Free PMC article.
-
Interferons and epigenetic mechanisms in training, priming and tolerance of monocytes and hematopoietic progenitors.Immunol Rev. 2024 May;323(1):257-275. doi: 10.1111/imr.13330. Epub 2024 Apr 3. Immunol Rev. 2024. PMID: 38567833 Free PMC article. Review.
References
-
- World Health Organization. World Malaria Report 2021. World Health Organization; 2021.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

