A biophysical threshold for biofilm formation
- PMID: 35642782
- PMCID: PMC9302973
- DOI: 10.7554/eLife.76380
A biophysical threshold for biofilm formation
Abstract
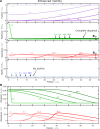
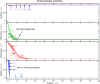
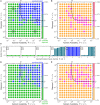
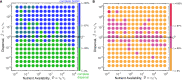
Bacteria are ubiquitous in our daily lives, either as motile planktonic cells or as immobilized surface-attached biofilms. These different phenotypic states play key roles in agriculture, environment, industry, and medicine; hence, it is critically important to be able to predict the conditions under which bacteria transition from one state to the other. Unfortunately, these transitions depend on a dizzyingly complex array of factors that are determined by the intrinsic properties of the individual cells as well as those of their surrounding environments, and are thus challenging to describe. To address this issue, here, we develop a generally-applicable biophysical model of the interplay between motility-mediated dispersal and biofilm formation under positive quorum sensing control. Using this model, we establish a universal rule predicting how the onset and extent of biofilm formation depend collectively on cell concentration and motility, nutrient diffusion and consumption, chemotactic sensing, and autoinducer production. Our work thus provides a key step toward quantitatively predicting and controlling biofilm formation in diverse and complex settings.
Keywords: biofilm; chemotaxis; computational biology; dispersal; motility; none; physics of living systems; quorum sensing; systems biology.
© 2022, Moore-Ott et al.
Conflict of interest statement
JM, SC, DA, TB, SD No competing interests declared
Figures

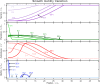
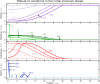
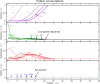
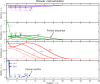
Comment in
- doi: 10.7554/eLife.80891
References
-
- Abdel-Aziz S. Bacterial Biofilm: Dispersal and Inhibition Strategies. Scholarena Journal of Biotechnology. 2014;10:105. doi: 10.18875/2375-6713.1.105. - DOI
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

