Recent developments in the structural characterisation of the IR and IGF1R: implications for the design of IR-IGF1R hybrid receptor modulators
- PMID: 35647546
- PMCID: PMC9020618
- DOI: 10.1039/d1md00300c
Recent developments in the structural characterisation of the IR and IGF1R: implications for the design of IR-IGF1R hybrid receptor modulators
Abstract
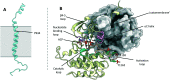
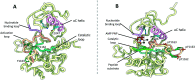
The insulin receptor (IR) and insulin-like growth factor 1 receptor (IGF1R) are dimeric disulfide-linked receptor tyrosine kinases, whose actions regulate metabolic and mitogenic signalling pathways inside the cell. It is well documented that in tissues co-expressing the IR and IGF1R, their respective monomers can heterodimerise to form IR-IGF1R hybrid receptors. Increased populations of the IR-IGF1R hybrid receptors are associated with several disease states, including type 2 diabetes and cancer. Recently, progress in the structural biology of IR and IGF1R has given insights into their structure-function relationships and mechanism of action. However, challenges in isolating IR-IGF1R hybrid receptors mean that their structural properties remain relatively unexplored. This review discusses the advances in the structural understanding of the IR and IGF1R, and how these discoveries can inform the design of small-molecule modulators of the IR-IGF1R hybrid receptors to understand their role in cell biology.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

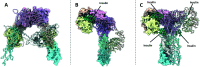
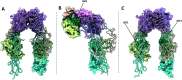



Similar articles
-
Molecular basis for the role of disulfide-linked αCTs in the activation of insulin-like growth factor 1 receptor and insulin receptor.Elife. 2022 Nov 22;11:e81286. doi: 10.7554/eLife.81286. Elife. 2022. PMID: 36413010 Free PMC article.
-
Down-regulation of type I insulin-like growth factor receptor increases sensitivity of breast cancer cells to insulin.Cancer Res. 2007 Jan 1;67(1):391-7. doi: 10.1158/0008-5472.CAN-06-1712. Cancer Res. 2007. PMID: 17210722
-
Targeting non-small cell lung cancer cells by dual inhibition of the insulin receptor and the insulin-like growth factor-1 receptor.PLoS One. 2013 Jun 24;8(6):e66963. doi: 10.1371/journal.pone.0066963. Print 2013. PLoS One. 2013. PMID: 23826179 Free PMC article.
-
Insulin/Insulin-like growth factors in cancer: new roles for the aryl hydrocarbon receptor, tumor resistance mechanisms, and new blocking strategies.Front Endocrinol (Lausanne). 2015 Feb 2;6:12. doi: 10.3389/fendo.2015.00012. eCollection 2015. Front Endocrinol (Lausanne). 2015. PMID: 25699021 Free PMC article. Review.
-
Cell cycle control by the insulin-like growth factor signal: at the crossroad between cell growth and mitotic regulation.Cell Cycle. 2023 Jan;22(1):1-37. doi: 10.1080/15384101.2022.2108117. Epub 2022 Aug 25. Cell Cycle. 2023. PMID: 36005738 Free PMC article. Review.
Cited by
-
Mass Spectrometry and Pharmacological Approaches to Measuring Cooption and Reciprocal Activation of Receptor Tyrosine Kinases.Proteomes. 2023 Jun 2;11(2):20. doi: 10.3390/proteomes11020020. Proteomes. 2023. PMID: 37368466 Free PMC article.
-
Insulin receptor Arg717 and IGF-1 receptor Arg704 play a key role in ligand binding and in receptor activation.Open Biol. 2023 Nov;13(11):230142. doi: 10.1098/rsob.230142. Epub 2023 Nov 8. Open Biol. 2023. PMID: 37935358 Free PMC article.
-
Identifying antisense oligonucleotides for targeted inhibition of insulin receptor isoform A.Front Oncol. 2025 Apr 15;15:1563985. doi: 10.3389/fonc.2025.1563985. eCollection 2025. Front Oncol. 2025. PMID: 40303997 Free PMC article.
-
IGF1R/ARRB1 Mediated Regulation of ERK and cAMP Pathways in Response to Aβ Unfolds Novel Therapeutic Avenue in Alzheimer's Disease.Mol Neurobiol. 2025 Jun;62(6):8065-8083. doi: 10.1007/s12035-025-04735-6. Epub 2025 Feb 19. Mol Neurobiol. 2025. PMID: 39969678 Free PMC article.
-
Single-chain insulin analogs threaded by the insulin receptor αCT domain.Biophys J. 2022 Nov 1;121(21):4063-4077. doi: 10.1016/j.bpj.2022.09.038. Epub 2022 Sep 30. Biophys J. 2022. PMID: 36181268 Free PMC article.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Chemical Information
Molecular Biology Databases

