Prodrugs of pyrophosphates and bisphosphonates: disguising phosphorus oxyanions
- PMID: 35647550
- PMCID: PMC9020613
- DOI: 10.1039/d1md00297j
Prodrugs of pyrophosphates and bisphosphonates: disguising phosphorus oxyanions
Abstract
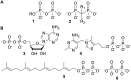
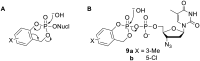
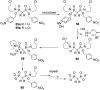
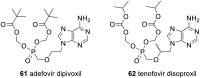
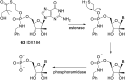
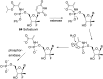
Pyrophosphates have important functions in living systems and thus pyrophosphate-containing molecules and their more stable bisphosphonate analogues have the potential to be used as drugs for treating many diseases including cancer and viral infections. Both pyrophosphates and bisphosphonates are polyanionic at physiological pH and, whilst this is essential for their biological activity, it also limits their use as therapeutic agents. In particular, the high negative charge density of these compounds prohibits cell entry other than by endocytosis, prevents transcellular oral absorption and causes sequestration to bone. Therefore, prodrug strategies have been developed to temporarily disguise the charges of these compounds. This review examines the various systems that have been used to mask the phosphorus-containing moieties of pyrophosphates and bisphosphonates and also illustrates the utility of such prodrugs.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures
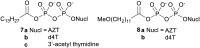
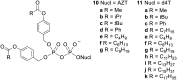




Similar articles
-
Bisphosphonates: from the laboratory to the clinic and back again.Bone. 1999 Jul;25(1):97-106. doi: 10.1016/s8756-3282(99)00116-7. Bone. 1999. PMID: 10423031 Review.
-
Bisphosphonate prodrugs.Curr Med Chem. 2002 Jun;9(12):1201-8. doi: 10.2174/0929867023369998. Curr Med Chem. 2002. PMID: 12052172 Review.
-
Hydroxy- and Amino-Phosphonates and -Bisphosphonates: Synthetic Methods and Their Biological Applications.Front Chem. 2022 Jun 1;10:890696. doi: 10.3389/fchem.2022.890696. eCollection 2022. Front Chem. 2022. PMID: 35721002 Free PMC article. Review.
-
Bisphosphonates: the first 40 years.Bone. 2011 Jul;49(1):2-19. doi: 10.1016/j.bone.2011.04.022. Epub 2011 May 1. Bone. 2011. PMID: 21555003 Review.
-
Anti-Tumor Activity and Immunotherapeutic Potential of a Bisphosphonate Prodrug.Sci Rep. 2017 Jul 20;7(1):5987. doi: 10.1038/s41598-017-05553-0. Sci Rep. 2017. PMID: 28729550 Free PMC article.
Cited by
-
The key phosphorus moieties in drug design: antimicrobials and beyond.Future Med Chem. 2024;16(23):2455-2458. doi: 10.1080/17568919.2024.2423602. Epub 2024 Nov 19. Future Med Chem. 2024. PMID: 39560019 No abstract available.
-
Triazole-Based Thiamine Analogues as Inhibitors of Thiamine Pyrophosphate-Dependent Enzymes: 1,3-Dicarboxylate for Metal Binding.ACS Omega. 2024 Sep 30;9(41):42245-42252. doi: 10.1021/acsomega.4c04594. eCollection 2024 Oct 15. ACS Omega. 2024. PMID: 39431096 Free PMC article.
-
Inhibition of Thiamine Diphosphate-Dependent Enzymes by Triazole-Based Thiamine Analogues.ACS Med Chem Lett. 2023 Apr 11;14(5):621-628. doi: 10.1021/acsmedchemlett.3c00047. eCollection 2023 May 11. ACS Med Chem Lett. 2023. PMID: 37197459 Free PMC article.
-
Cytotoxicity of phosphoramidate, bis-amidate and cycloSal prodrug metabolites against tumour and normal cells.RSC Med Chem. 2024 Apr 18;15(6):1973-1981. doi: 10.1039/d4md00115j. eCollection 2024 Jun 19. RSC Med Chem. 2024. PMID: 38903945 Free PMC article.
References
-
- Berg J. M., Tymoczko J. L., Gatto G. and Stryer L., Biochemistry, W.H. Freeman and Company, New York, 8th edn, 2015
Publication types
LinkOut - more resources
Full Text Sources

