Cobalt-Catalyzed Cyclization of Unsaturated N-Acyl Sulfonamides: a Diverted Mukaiyama Hydration Reaction
- PMID: 35647594
- PMCID: PMC9131372
- DOI: 10.1021/jacsau.2c00186
Cobalt-Catalyzed Cyclization of Unsaturated N-Acyl Sulfonamides: a Diverted Mukaiyama Hydration Reaction
Abstract
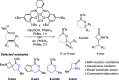
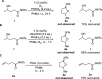
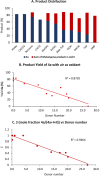
The cycloisomerization of β-, γ-, and δ-unsaturated N-acyl sulfonamides to N-sulfonyl lactams and imidates is reported. This transformation is effected in the presence of a CoIII(salen) catalyst using t-BuOOH or air as the oxidant. The method shows good functional group tolerance (alkyl, aryl, heteroaryl, ether, N-Boc) and furnishes an underexplored class of cyclic building blocks. The strong solvent dependence of the transformation is investigated, and the synthetic versatility of the N-sulfonyl imidate product class is highlighted.
© 2022 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures





Similar articles
-
Cobalt-Catalyzed Aerobic Aminocyclization of Unsaturated Amides for the Synthesis of Functionalized γ- and δ-Lactams.Org Lett. 2023 Sep 1;25(34):6380-6384. doi: 10.1021/acs.orglett.3c02390. Epub 2023 Aug 23. Org Lett. 2023. PMID: 37610083 Free PMC article.
-
Cobalt-Catalyzed Hartung-Mukaiyama Cyclization of γ-Hydroxy Olefins: Stereocontrolled Synthesis of the Tetrahydrofuran Moiety of Amphidinolide N.J Org Chem. 2021 Apr 16;86(8):5584-5615. doi: 10.1021/acs.joc.1c00085. Epub 2021 Mar 26. J Org Chem. 2021. PMID: 33769047
-
Ru-catalyzed cyclocarbonylation of alpha- and beta-allenic sulfonamides: synthesis of gamma- and delta-unsaturated lactams.Org Lett. 2001 Sep 6;3(18):2851-3. doi: 10.1021/ol016281c. Org Lett. 2001. PMID: 11529773
-
Studies on Catalytic Activation of Olefins Using Cobalt Complex.Chem Pharm Bull (Tokyo). 2018;66(4):339-346. doi: 10.1248/cpb.c17-01006. Chem Pharm Bull (Tokyo). 2018. PMID: 29607898 Review.
-
Imidates: an emerging synthon for N-heterocycles.Org Biomol Chem. 2019 Nov 27;17(46):9829-9843. doi: 10.1039/c9ob01899a. Org Biomol Chem. 2019. PMID: 31720673 Review.
Cited by
-
Cobalt-Catalyzed Aerobic Aminocyclization of Unsaturated Amides for the Synthesis of Functionalized γ- and δ-Lactams.Org Lett. 2023 Sep 1;25(34):6380-6384. doi: 10.1021/acs.orglett.3c02390. Epub 2023 Aug 23. Org Lett. 2023. PMID: 37610083 Free PMC article.
-
Intermolecular Organophotocatalytic Cyclopropanation of Unactivated Olefins.J Am Chem Soc. 2023 Jan 18;145(2):774-780. doi: 10.1021/jacs.2c11680. Epub 2023 Jan 6. J Am Chem Soc. 2023. PMID: 36607827 Free PMC article.
-
Organophotocatalytic carbo-heterofunctionalization of unactivated olefins with pendant nucleophiles.Chem Sci. 2023 Jun 14;14(26):7256-7261. doi: 10.1039/d3sc02250a. eCollection 2023 Jul 5. Chem Sci. 2023. PMID: 37416720 Free PMC article.
-
Iron-Mediated Photochemical Anti-Markovnikov Hydroazidation of Unactivated Olefins.J Am Chem Soc. 2023 Oct 18;145(41):22347-22353. doi: 10.1021/jacs.3c09122. Epub 2023 Oct 9. J Am Chem Soc. 2023. PMID: 37811819 Free PMC article.
References
-
- Crossley S. W. M.; Obradors C.; Martinez R. M.; Shenvi R. A. Mn-, Fe-, and Co-Catalyzed Radical Hydrofunctionalizations of Olefins. Chem. Rev. 2016, 116, 8912–9000. 10.1021/acs.chemrev.6b00334. - DOI - PMC - PubMed
- Kyne S. H.; Lefevre G.; Ollivier C.; Petit M.; Ramis Cladera V. A.; Fensterbank L. Iron and cobalt catalysis: new perspectives in synthetic radical chemistry. Chem. Soc. Rev. 2020, 49, 8501–8542. 10.1039/D0CS00969E. - DOI - PubMed
-
- Ludwig J. R.; Schindler C. S. Catalyst: Sustainable Catalysis. Chem. 2017, 2, 313–316. 10.1016/j.chempr.2017.02.014. - DOI
-
- Lee N.-H.; Byun J.-C.; Baik J.-S.; Han C.-H.; Han S.-b. Development of Mn(III)(Schiff Base) Complexes for the Catalyst of Olefin Oxygenation to Alcohols in the Presence of NaBH4. Bull. Korean Chem. Soc. 2002, 23, 1365–1366. 10.5012/bkcs.2002.23.10.1365. - DOI
-
- Matsushita Y.-I.; Sugamoto K.; Nakama T.; Matsui T. Synthesis of γ-hydroperoxy-α,β-unsaturated carbonyl compounds from α,β,γ,δ-unsaturated carbonyl compounds by cobalt(II) porphyrin-catalysed hydroperoxygenation. J. Chem. Soc., Chem. Commun. 1995, 567–568. 10.1039/C39950000567. - DOI
- Sugamoto K.; Matsushita Y.-I.; Matsui T. Direct hydroperoxygenation of conjugated olefins catalyzed by cobalt(II) porphyrin. J. Chem. Soc., Perkin Trans. 1 1998, 3989–3998. 10.1039/a805888a. - DOI
- Tokuyasu T.; Kunikawa S.; Masuyama A.; Nojima M. Co(III)–Alkyl Complex- and Co(III)–Alkylperoxo Complex-Catalyzed Triethylsilylperoxidation of Alkenes with Molecular Oxygen and Triethylsilane. Org. Lett. 2002, 4, 3595–3598. 10.1021/ol0201299. - DOI - PubMed
- O’Neill P. M.; Hindley S.; Pugh M. D.; Davies J.; Bray P. G.; Park B. K.; Kapu D. S.; Ward S. A.; Stocks P. A. Co(thd)2: a superior catalyst for aerobic epoxidation and hydroperoxysilylation of unactivated alkenes: application to the synthesis of spiro-1,2,4-trioxanes. Tetrahedron Lett. 2003, 44, 8135–8138. 10.1016/j.tetlet.2003.09.033. - DOI
- Tokuyasu T.; Kunikawa S.; McCullough K. J.; Masuyama A.; Nojima M. Synthesis of Cyclic Peroxides by Chemo- and Regioselective Peroxidation of Dienes with Co(II)/O2/Et3SiH. J. Org. Chem. 2005, 70, 251–260. 10.1021/jo048359j. - DOI - PubMed
- Hurlocker B.; Miner M. R.; Woerpel K. A. Synthesis of silyl monoperoxyketals by regioselective cobalt-catalyzed peroxidation of silyl enol ethers: application to the synthesis of 1,2-dioxolanes. Org. Lett. 2014, 16, 4280–4283. 10.1021/ol5020015. - DOI - PubMed
-
- Zombeck A.; Hamilton D. E.; Drago R. S. Novel catalytic oxidations of terminal olefins by cobalt(II)-Schiff base complexes. J. Am. Chem. Soc. 1982, 104, 6782–6784. 10.1021/ja00388a051. - DOI
LinkOut - more resources
Full Text Sources
