Crystal structure of an extracellular superoxide dismutase from Onchocerca volvulus and implications for parasite-specific drug development
- PMID: 35647680
- PMCID: PMC9158661
- DOI: 10.1107/S2053230X22005350
Crystal structure of an extracellular superoxide dismutase from Onchocerca volvulus and implications for parasite-specific drug development
Abstract
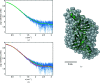
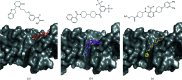
Superoxide dismutases (SODs) are metalloproteins that are responsible for the dismutation of superoxide anion radicals. SODs are consequently protective against oxidative damage to cellular components. Among other protective mechanisms, the filarial parasite Onchocerca volvulus has a well developed defense system to scavenge toxic free radicals using SODs during migration and sojourning of the microfilariae and adult worms in the human body. O. volvulus is responsible for the neglected disease onchocerciasis or `river blindness'. In the present study, an extracellular Cu/Zn-SOD from O. volvulus (OvEC-SOD) was cloned, purified and crystallized to obtain structural insight into an attractive drug target with the potential to combat onchocerciasis. The recombinant OvEC-SOD forms a dimer and the protein structure was solved and refined to 1.55 Å resolution by X-ray crystallography. Interestingly, a sulfate ion supports the coordination of the conserved copper ion. The overall protein shape was verified by small-angle X-ray scattering. The enzyme shows a different surface charge distribution and different termini when compared with the homologous human SOD. A distinct hydrophobic cleft to which both protomers of the dimer contribute was utilized for a docking approach with compounds that have previously been identified as SOD inhibitors to highlight the potential for individual structure-based drug development.
Keywords: Cu/Zn superoxide dismutases; Onchocerca volvulus; X-ray crystallography; docking; drug targets; metal ion coordination; parasites.
open access.
Figures

References
-
- Ascone, I., Castañer, R., Tarricone, C., Bolognesi, M., Stroppolo, M. E. & Desideri, A. (1997). Biochem. Biophys. Res. Commun. 241, 119–121. - PubMed
-
- Assfalg, M., Banci, L., Bertini, I., Turano, P. & Vasos, P. R. (2003). J. Mol. Biol. 330, 145–158. - PubMed
-
- Banci, L., Bertini, I., Hallewell, R. A., Luchinat, C. & Viezzoli, M. S. (1989). Eur. J. Biochem. 184, 125–129. - PubMed
-
- Basáñez, M.-G., Pion, S. D., Boakes, E., Filipe, J. A., Churcher, T. S. & Boussinesq, M. (2008). Lancet Infect. Dis. 8, 310–322. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

