Deep phenotyping for precision medicine in Parkinson's disease
- PMID: 35647913
- PMCID: PMC9178512
- DOI: 10.1242/dmm.049376
Deep phenotyping for precision medicine in Parkinson's disease
Abstract
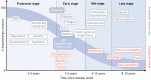
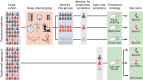
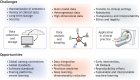
A major challenge in medical genomics is to understand why individuals with the same disorder have different clinical symptoms and why those who carry the same mutation may be affected by different disorders. In every complex disorder, identifying the contribution of different genetic and non-genetic risk factors is a key obstacle to understanding disease mechanisms. Genetic studies rely on precise phenotypes and are unable to uncover the genetic contributions to a disorder when phenotypes are imprecise. To address this challenge, deeply phenotyped cohorts have been developed for which detailed, fine-grained data have been collected. These cohorts help us to investigate the underlying biological pathways and risk factors to identify treatment targets, and thus to advance precision medicine. The neurodegenerative disorder Parkinson's disease has a diverse phenotypical presentation and modest heritability, and its underlying disease mechanisms are still being debated. As such, considerable efforts have been made to develop deeply phenotyped cohorts for this disorder. Here, we focus on Parkinson's disease and explore how deep phenotyping can help address the challenges raised by genetic and phenotypic heterogeneity. We also discuss recent methods for data collection and computation, as well as methodological challenges that have to be overcome.
Keywords: Genetics; Phenotyping; Precision medicine.
© 2022. Published by The Company of Biologists Ltd.
Conflict of interest statement
Competing interests The authors declare no competing or financial interests.
Figures




References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

