Sex-Biased Control of Inflammation and Metabolism by a Mitochondrial Nod-Like Receptor
- PMID: 35651602
- PMCID: PMC9150262
- DOI: 10.3389/fimmu.2022.882867
Sex-Biased Control of Inflammation and Metabolism by a Mitochondrial Nod-Like Receptor
Abstract
Mitochondria regulate steroid hormone synthesis, and in turn sex hormones regulate mitochondrial function for maintaining cellular homeostasis and controlling inflammation. This crosstalk can explain sex differences observed in several pathologies such as in metabolic or inflammatory disorders. Nod-like receptor X1 (NLRX1) is a mitochondria-associated innate receptor that could modulate metabolic functions and attenuates inflammatory responses. Here, we showed that in an infectious model with the human protozoan parasite, Leishmania guyanensis, NLRX1 attenuated inflammation in females but not in male mice. Analysis of infected female and male bone marrow derived macrophages showed both sex- and genotype-specific differences in both inflammatory and metabolic profiles with increased type I interferon production, mitochondrial respiration, and glycolytic rate in Nlrx1-deficient female BMDMs in comparison to wild-type cells, while no differences were observed between males. Transcriptomics of female and male BMDMs revealed an altered steroid hormone signaling in Nlrx1-deficient cells, and a "masculinization" of Nlrx1-deficient female BMDMs. Thus, our findings suggest that NLRX1 prevents uncontrolled inflammation and metabolism in females and therefore may contribute to the sex differences observed in infectious and inflammatory diseases.
Keywords: inflammation; innate immunity; metabolism; nod-like receptor X1; sex.
Copyright © 2022 Snäkä, Bekkar, Desponds, Prével, Claudinot, Isorce, Teixeira, Grasset, Xenarios, Lopez-Mejia, Fajas and Fasel.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
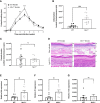

 represents modules enriched in GO terms associated with sex hormone signaling. Graphs are presented as mean +- SEM and significance was tested by two-way ANOVA with multiple comparisons (A-F). ns = non-significant, *p ≤ 0.05, **p ≤ 0.01, ***p ≤ 0.001.
represents modules enriched in GO terms associated with sex hormone signaling. Graphs are presented as mean +- SEM and significance was tested by two-way ANOVA with multiple comparisons (A-F). ns = non-significant, *p ≤ 0.05, **p ≤ 0.01, ***p ≤ 0.001.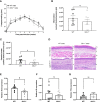

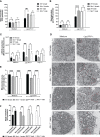

References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

