Advancing homogeneous catalysis for parahydrogen-derived hyperpolarisation and its NMR applications
- PMID: 35655870
- PMCID: PMC9067625
- DOI: 10.1039/d2sc00737a
Advancing homogeneous catalysis for parahydrogen-derived hyperpolarisation and its NMR applications
Abstract
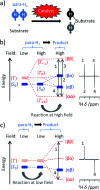
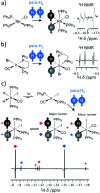
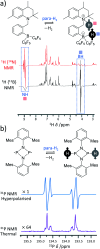
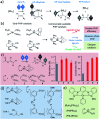
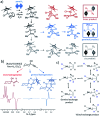
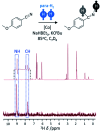
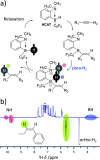
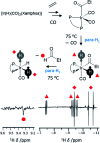
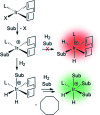
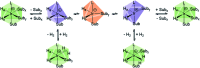
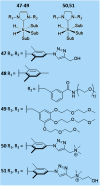
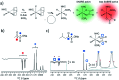
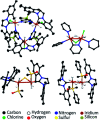
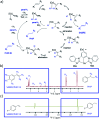
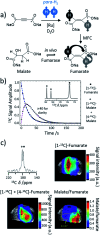
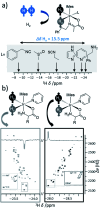
Parahydrogen-induced polarisation (PHIP) is a nuclear spin hyperpolarisation technique employed to enhance NMR signals for a wide range of molecules. This is achieved by exploiting the chemical reactions of parahydrogen (para-H2), the spin-0 isomer of H2. These reactions break the molecular symmetry of para-H2 in a way that can produce dramatically enhanced NMR signals for reaction products, and are usually catalysed by a transition metal complex. In this review, we discuss recent advances in novel homogeneous catalysts that can produce hyperpolarised products upon reaction with para-H2. We also discuss hyperpolarisation attained in reversible reactions (termed signal amplification by reversible exchange, SABRE) and focus on catalyst developments in recent years that have allowed hyperpolarisation of a wider range of target molecules. In particular, recent examples of novel ruthenium catalysts for trans and geminal hydrogenation, metal-free catalysts, iridium sulfoxide-containing SABRE systems, and cobalt complexes for PHIP and SABRE are reviewed. Advances in this catalysis have expanded the types of molecules amenable to hyperpolarisation using PHIP and SABRE, and their applications in NMR reaction monitoring, mechanistic elucidation, biomedical imaging, and many other areas, are increasing.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures
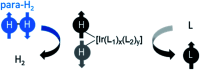



Similar articles
-
Unconventional Parahydrogen-Induced Hyperpolarization Effects in Chemistry and Catalysis: From Photoreactions to Enzymes.ACS Catal. 2025 Apr 4;15(8):6386-6409. doi: 10.1021/acscatal.4c07870. eCollection 2025 Apr 18. ACS Catal. 2025. PMID: 40270879 Free PMC article. Review.
-
Quantification of hyperpolarisation efficiency in SABRE and SABRE-Relay enhanced NMR spectroscopy.Phys Chem Chem Phys. 2018 Nov 7;20(41):26362-26371. doi: 10.1039/c8cp05473h. Epub 2018 Oct 10. Phys Chem Chem Phys. 2018. PMID: 30303501 Free PMC article.
-
Developments and advances concerning the hyperpolarisation technique SABRE.Magn Reson Chem. 2015 Oct;53(10):789-800. doi: 10.1002/mrc.4280. Epub 2015 Aug 12. Magn Reson Chem. 2015. PMID: 26264565
-
Magnetic field dependence of the para-ortho conversion rate of molecular hydrogen in SABRE experiments.J Magn Reson. 2024 Mar;360:107630. doi: 10.1016/j.jmr.2024.107630. Epub 2024 Feb 15. J Magn Reson. 2024. PMID: 38364339
-
Hyperpolarised benchtop NMR spectroscopy for analytical applications.Prog Nucl Magn Reson Spectrosc. 2024 Nov-Dec;144-145:153-178. doi: 10.1016/j.pnmrs.2024.10.001. Epub 2024 Oct 22. Prog Nucl Magn Reson Spectrosc. 2024. PMID: 39645349 Review.
Cited by
-
Metal-Mediated Catalytic Polarization Transfer from para Hydrogen to 3,5-Dihalogenated Pyridines.ACS Catal. 2024 Jan 5;14(2):994-1004. doi: 10.1021/acscatal.3c05378. eCollection 2024 Jan 19. ACS Catal. 2024. PMID: 38269038 Free PMC article.
-
Enhancing the NMR signals of plant oil components using hyperpolarisation relayed via proton exchange.Chem Sci. 2023 Aug 29;14(36):9843-9853. doi: 10.1039/d3sc03078d. eCollection 2023 Sep 20. Chem Sci. 2023. PMID: 37736655 Free PMC article.
-
Unconventional Parahydrogen-Induced Hyperpolarization Effects in Chemistry and Catalysis: From Photoreactions to Enzymes.ACS Catal. 2025 Apr 4;15(8):6386-6409. doi: 10.1021/acscatal.4c07870. eCollection 2025 Apr 18. ACS Catal. 2025. PMID: 40270879 Free PMC article. Review.
-
Base-Promoted Iridium-Catalyzed Deuteration and C-H Bond Activation of N-Heterocycles.J Org Chem. 2025 Jun 20;90(24):8080-8089. doi: 10.1021/acs.joc.5c00174. Epub 2025 Jun 5. J Org Chem. 2025. PMID: 40471654 Free PMC article.
-
Spin Hyperpolarization in Modern Magnetic Resonance.Chem Rev. 2023 Feb 22;123(4):1417-1551. doi: 10.1021/acs.chemrev.2c00534. Epub 2023 Jan 26. Chem Rev. 2023. PMID: 36701528 Free PMC article. Review.
References
-
- Keeler J., Understanding NMR Spectroscopy, John Wiley & Sons, Chichester, 2010
-
- Hyperpolarization Methods in NMR Spectroscopy, Topics in Current Chemistry, ed. L. T. Kuhn, Springer, Berlin, 2013, vol. 338
-
- Ardenkjaer-Larsen J. Boebinger G. S. Comment A. Duckett S. Edison A. S. Engelke F. Griesinger C. Griffin R. G. Hilty C. Maeda H. Parigi G. Prisner T. Ravera E. van Bentum J. Vega S. Webb A. Luchinat C. Schwalbe H. Frydman L. Angew. Chem., Int. Ed. 2015;54:9162–9185. doi: 10.1002/anie.201410653. - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

