The Homeostasis of Cartilage Matrix Remodeling and the Regulation of Volume-Sensitive Ion Channel
- PMID: 35656105
- PMCID: PMC9116913
- DOI: 10.14336/AD.2021.1122
The Homeostasis of Cartilage Matrix Remodeling and the Regulation of Volume-Sensitive Ion Channel
Abstract
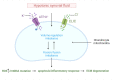
Degenerative joint diseases of the hips and knees are common and are accompanied by severe pain and movement disorders. At the microscopic level, the main characteristics of osteoarthritis are the continuous destruction and degeneration of cartilage, increased cartilage extracellular matrix catabolism, decreased anabolism, increased synovial fluid, and decreased osmotic pressure. Cell volume stability is mainly regulated by ion channels, many of which are expressed in chondrocytes. These ion channels are closely related to pain regulation, volume regulation, the inflammatory response, cell proliferation, apoptosis, and cell differentiation. In this review, we focus on the important role of volume control-related ion channels in cartilage matrix remodeling and summarize current views. In addition, the potential mechanism of the volume-sensitive anion channel LRRC8A in the early occurrence of osteoarthritis is discussed.
Keywords: cell volume regulation; chondrocytes; ion channel; osteoarthritis.
Copyright: © 2022 Deng et al.
Conflict of interest statement
Competing interests The authors declare that they have no competing interests.
Figures




Similar articles
-
Chloride Channel and Inflammation-Mediated Pathogenesis of Osteoarthritis.J Inflamm Res. 2022 Feb 11;15:953-964. doi: 10.2147/JIR.S350432. eCollection 2022. J Inflamm Res. 2022. PMID: 35177922 Free PMC article. Review.
-
Activation of a chondrocyte volume-sensitive Cl(-) conductance prior to macroscopic cartilage lesion formation in the rabbit knee anterior cruciate ligament transection osteoarthritis model.Osteoarthritis Cartilage. 2016 Oct;24(10):1786-1794. doi: 10.1016/j.joca.2016.05.019. Epub 2016 Jun 4. Osteoarthritis Cartilage. 2016. PMID: 27266646 Free PMC article.
-
Ion transport in chondrocytes: membrane transporters involved in intracellular ion homeostasis and the regulation of cell volume, free [Ca2+] and pH.Histol Histopathol. 1998 Jul;13(3):893-910. doi: 10.14670/HH-13.893. Histol Histopathol. 1998. PMID: 9690144 Review.
-
Ion channel expression and function in normal and osteoarthritic human synovial fluid progenitor cells.Channels (Austin). 2016;10(2):148-57. doi: 10.1080/19336950.2015.1116652. Epub 2015 Dec 2. Channels (Austin). 2016. PMID: 26632350 Free PMC article.
-
Chondropenia: current concept review.Musculoskelet Surg. 2015 Dec;99(3):189-200. doi: 10.1007/s12306-015-0377-9. Epub 2015 Jun 13. Musculoskelet Surg. 2015. PMID: 26068954 Review.
Cited by
-
TMEM16 proteins: Ca2+‑activated chloride channels and phospholipid scramblases as potential drug targets (Review).Int J Mol Med. 2024 Oct;54(4):81. doi: 10.3892/ijmm.2024.5405. Epub 2024 Aug 2. Int J Mol Med. 2024. PMID: 39092585 Free PMC article. Review.
-
Osteoarthritis, adipokines and the translational research potential in small animal patients.Front Vet Sci. 2024 May 20;11:1193702. doi: 10.3389/fvets.2024.1193702. eCollection 2024. Front Vet Sci. 2024. PMID: 38831954 Free PMC article. Review.
-
Bibliometric analysis of chondrocyte apoptosis in knee osteoarthritis.Medicine (Baltimore). 2024 Oct 4;103(40):e40000. doi: 10.1097/MD.0000000000040000. Medicine (Baltimore). 2024. PMID: 39465698 Free PMC article.
-
Single‑cell sequencing, genetics, and epigenetics reveal mesenchymal stem cell senescence in osteoarthritis (Review).Int J Mol Med. 2024 Jan;53(1):2. doi: 10.3892/ijmm.2023.5326. Epub 2023 Nov 8. Int J Mol Med. 2024. PMID: 37937669 Free PMC article. Review.
-
Apoptosis Regulation in Osteoarthritis and the Influence of Lipid Interactions.Int J Mol Sci. 2023 Aug 22;24(17):13028. doi: 10.3390/ijms241713028. Int J Mol Sci. 2023. PMID: 37685835 Free PMC article. Review.
References
-
- Kloppenburg M, Berenbaum F (2020). Osteoarthritis year in review 2019: epidemiology and therapy. Osteoarthritis Cartilage, 28:242-248. - PubMed
-
- Glyn-Jones S, Palmer AJR, Agricola R, Price AJ, Vincent TL, Weinans H, et al.. (2015). Osteoarthritis. Lancet, 386:376-387. - PubMed
-
- Teyhen DS (2017). Running and Osteoarthritis Does Recreational or Competitive Running Increase the Risk? J Orthop Sports Phys Ther, 47:391-391. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
