Low Dose Betahistine in Combination With Selegiline Increases Cochlear Blood Flow in Guinea Pigs
- PMID: 35656811
- PMCID: PMC10108341
- DOI: 10.1177/00034894221098803
Low Dose Betahistine in Combination With Selegiline Increases Cochlear Blood Flow in Guinea Pigs
Abstract
Objective: Betahistine is frequently used in the pharmacotherapy for Menière's Disease (MD). Little is known about its mode of action and prescribed dosages vary. While betahistine had an increasing effect on cochlear microcirculation in earlier studies, low dose betahistine of 0.01 mg/kg bw or less was not able to effect this. Selegiline inhibits monoaminooxidase B and therefore potentially the breakdown of betahistine. The goal of this study was to examine whether the addition of selegiline to low dose betahistine leads to increased cochlear blood flow.
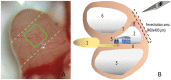
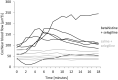
Methods: Twelve Dunkin-Hartley guinea pigs were anesthetized, the cochlea was exposed and a window opened to the stria vascularis. Blood plasma was visualized by injecting fluoresceinisothiocyanate-dextrane and vessel diameter and erythrocyte velocity were evaluated over 20 minutes. One group received low dose betahistine (0.01 mg/kg bw) and selegiline (1 mg/kg bw) i.v. while the other group received only selegiline (1 mg/kg bw) and saline (0.9% NaCl) as placebo i.v.
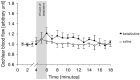
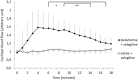
Results: Cochlear microcirculation increased significantly (P < .001) in guinea pigs treated with low dose betahistine combined with selegiline by up to 58.3 ± 38.7% above baseline over a period of up to 11 minutes. In one guinea pig, the increase was 104.6%. Treatment with Selegiline alone did not affect microcirculation significantly.
Conclusions: Low dose betahistine increased cochlear microcirculation significantly when combined with selegiline. This should be investigated in further studies regarding dose-effect relation in comparison to betahistine alone. Side effects, in particular regarding circulation, should be considered carefully in view of the clinical applicability of a combination therapy in patients with MD.
Keywords: Menière’s disease; betahistine; cochlea; guinea pig; microcirculation; selegiline.
Conflict of interest statement
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JS received travel expenses and congress fees from MED EL GmbH, Innsbruck, Austria. The other authors declare that there is no conflict of interest.
Figures
Similar articles
-
Betahistine metabolites, aminoethylpyridine, and hydroxyethylpyridine increase cochlear blood flow in guinea pigs in vivo.Int J Audiol. 2014 Oct;53(10):753-9. doi: 10.3109/14992027.2014.917208. Epub 2014 Jul 11. Int J Audiol. 2014. PMID: 25014609
-
Betahistine exerts a dose-dependent effect on cochlear stria vascularis blood flow in guinea pigs in vivo.PLoS One. 2012;7(6):e39086. doi: 10.1371/journal.pone.0039086. Epub 2012 Jun 20. PLoS One. 2012. PMID: 22745706 Free PMC article.
-
Menière's disease: combined pharmacotherapy with betahistine and the MAO-B inhibitor selegiline-an observational study.J Neurol. 2018 Oct;265(Suppl 1):80-85. doi: 10.1007/s00415-018-8809-8. Epub 2018 Mar 12. J Neurol. 2018. Retraction in: J Neurol. 2020 Apr;267(4):1225. doi: 10.1007/s00415-020-09791-7. PMID: 29532287 Retracted.
-
Report from a Consensus Conference on the treatment of Ménière's disease with betahistine: rationale, methodology and results.Acta Otorhinolaryngol Ital. 2018 Oct;38(5):460-467. doi: 10.14639/0392-100X-2035. Acta Otorhinolaryngol Ital. 2018. PMID: 30498275 Free PMC article.
-
Betahistine in Ménière's Disease or Syndrome: A Systematic Review.Audiol Neurootol. 2022;27(1):1-33. doi: 10.1159/000515821. Epub 2021 Jul 7. Audiol Neurootol. 2022. PMID: 34233329
Cited by
-
Examination of betahistine bioavailability in combination with the monoamine oxidase B inhibitor, selegiline, in humans-a non-randomized, single-sequence, two-period titration, open label single-center phase 1 study (PK-BeST).Front Neurol. 2023 Oct 18;14:1271640. doi: 10.3389/fneur.2023.1271640. eCollection 2023. Front Neurol. 2023. PMID: 37920833 Free PMC article.
References
-
- Ferrara S, Dispenza F. Cause, pathogenesis, clinical manifestations and treatment of meniere’s disease and endolymphatic hydrops. In: Dispenza MF. (ed.) Sensorineural Hearing Loss: Pathophysiology, Diagnosis and Treatment. Nova Science Publishers, Inc; 2019; 217–231.
-
- Perez-Carpena P, Lopez-Escamez JA. Current understanding and clinical management of Meniere’s disease: a systematic review. Semin Neurol. 2020;40:138-150. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

