Effectiveness of mRNA vaccine boosters against infection with the SARS-CoV-2 omicron (B.1.1.529) variant in Spain: a nationwide cohort study
- PMID: 35658998
- PMCID: PMC9162477
- DOI: 10.1016/S1473-3099(22)00292-4
Effectiveness of mRNA vaccine boosters against infection with the SARS-CoV-2 omicron (B.1.1.529) variant in Spain: a nationwide cohort study
Abstract
Background: The omicron (B.1.1.529) variant of SARS-CoV-2 has increased capacity to elude immunity and cause breakthrough infections. The aim of this study was to estimate the effectiveness of mRNA-based vaccine boosters (third dose) against infection with the omicron variant by age, sex, time since complete vaccination, type of primary vaccine, and type of booster.
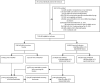
Methods: In this nationwide cohort study, we linked data from three nationwide population registries in Spain (Vaccination Registry, Laboratory Results Registry, and National Health System registry) to select community-dwelling individuals aged 40 years or older, who completed their primary vaccine schedule at least 3 months before the start of follow-up, and had not tested positive for SARS-CoV-2 since the start of the pandemic. On each day between Jan 3, and Feb 6, 2022, we matched individuals who received a booster mRNA vaccine and controls of the same sex, age group, postal code, type of vaccine, time since primary vaccination, and number of previous tests. We estimated risk of laboratory-confirmed SARS-CoV-2 infection using the Kaplan-Meier method and compared groups using risk ratios (RR) and risk differences. Vaccine effectiveness was calculated as one minus RR.
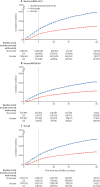
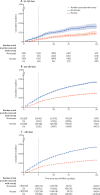
Findings: Between Jan 3, and Feb 6, 2022, 3 111 159 matched pairs were included in our study. Overall, the estimated effectiveness from day 7 to 34 after a booster was 51·3% (95% CI 50·2-52·4). Estimated effectiveness was 52·5% (51·3-53·7) for an mRNA-1273 booster and 46·2% (43·5-48·7) for a BNT162b2 booster. Effectiveness was 58·6% (55·5-61·6) if primary vaccination had been with ChAdOx1 nCoV-19 (Oxford-AstraZeneca), 55·3% (52·3-58·2) with mRNA-1273 (Moderna), 49·7% (48·3-51·1) with BNT162b2 (Pfizer-BioNTech), and 48·0% (42·5-53·7) with Ad26.COV2.S (Janssen). Estimated effectiveness was 43·6% (40·0-47·1) when the booster was administered between 151 days and 180 days after complete vaccination and 52·2% (51·0-53·3) if administered more than 180 days after primary scheduled completion.
Interpretation: Booster mRNA vaccine-doses were moderately effective in preventing infection with the omicron variant of SARS-CoV-2 for over a month after administration, which indicates their suitability as a strategy to limit the health effects of COVID-19 in periods of omicron variant domination. Estimated effectiveness was higher for mRNA-1273 compared with BNT162b2 and increased with time between completed primary vaccination and booster.
Funding: None.
Copyright © 2022 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Declaration of interests We declare no competing interest.
Figures
Comment in
-
Effectiveness of mRNA booster doses against the omicron variant.Lancet Infect Dis. 2022 Sep;22(9):1257-1258. doi: 10.1016/S1473-3099(22)00319-X. Epub 2022 Jun 2. Lancet Infect Dis. 2022. PMID: 35658999 Free PMC article. No abstract available.
References
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

