Cytotoxicity Assessment of Surface-Modified Magnesium Hydroxide Nanoparticles
- PMID: 35664586
- PMCID: PMC9161253
- DOI: 10.1021/acsomega.1c06515
Cytotoxicity Assessment of Surface-Modified Magnesium Hydroxide Nanoparticles
Abstract
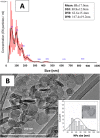
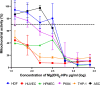
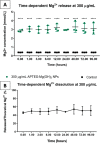
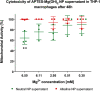
Magnesium-based nanoparticles have shown promise in regenerative therapies in orthopedics and the cardiovascular system. Here, we set out to assess the influence of differently functionalized Mg nanoparticles on the cellular players of wound healing, the first step in the process of tissue regeneration. First, we thoroughly addressed the physicochemical characteristics of magnesium hydroxide nanoparticles, which exhibited low colloidal stability and strong aggregation in cell culture media. To address this matter, magnesium hydroxide nanoparticles underwent surface functionalization by 3-aminopropyltriethoxysilane (APTES), resulting in excellent dispersible properties in ethanol and improved colloidal stability in physiological media. The latter was determined as a concentration- and time-dependent phenomenon. There were no significant effects on THP-1 macrophage viability up to 1.500 μg/mL APTES-coated magnesium hydroxide nanoparticles. Accordingly, increased media pH and Mg2+ concentration, the nanoparticles dissociation products, had no adverse effects on their viability and morphology. HDF, ASCs, and PK84 exhibited the highest, and HUVECs, HPMECs, and THP-1 cells the lowest resistance toward nanoparticle toxic effects. In conclusion, the indicated high magnesium hydroxide nanoparticles biocompatibility suggests them a potential drug delivery vehicle for treating diseases like fibrosis or cancer. If delivered in a targeted manner, cytotoxic nanoparticles could be considered a potential localized and specific prevention strategy for treating highly prevalent diseases like fibrosis or cancer. Looking toward the possible clinical applications, accurate interpretation of in vitro cellular responses is the keystone for the relevant prediction of subsequent in vivo biological effects.
© 2022 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures




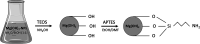
References
-
- Krishnamoorthy K.; Moon J. Y.; Hyun H. B.; Cho S. K.; Kim S.-J. Mechanistic investigation on the toxicity of MgO nanoparticles toward cancer cells. J. Mater. Chem. 2012, 22 (47), 24610–7. 10.1039/c2jm35087d. - DOI
LinkOut - more resources
Full Text Sources

