Alternative splicing encodes functional intracellular CD59 isoforms that mediate insulin secretion and are down-regulated in diabetic islets
- PMID: 35666870
- PMCID: PMC9214515
- DOI: 10.1073/pnas.2120083119
Alternative splicing encodes functional intracellular CD59 isoforms that mediate insulin secretion and are down-regulated in diabetic islets
Abstract
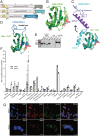
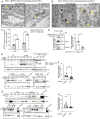
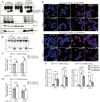
Human pancreatic islets highly express CD59, which is a glycosylphosphatidylinositol (GPI)-anchored cell-surface protein and is required for insulin secretion. How cell-surface CD59 could interact with intracellular exocytotic machinery has so far not been described. We now demonstrate the existence of CD59 splice variants in human pancreatic islets, which have unique C-terminal domains replacing the GPI-anchoring signal sequence. These isoforms are found in the cytosol of β-cells, interact with SNARE proteins VAMP2 and SNAP25, colocalize with insulin granules, and rescue insulin secretion in CD59-knockout (KO) cells. We therefore named these isoforms IRIS-1 and IRIS-2 (Isoforms Rescuing Insulin Secretion 1 and 2). Antibodies raised against each isoform revealed that expression of both IRIS-1 and IRIS-2 is significantly lower in islets isolated from human type 2 diabetes (T2D) patients, as compared to healthy controls. Further, glucotoxicity induced in primary, healthy human islets led to a significant decrease of IRIS-1 expression, suggesting that hyperglycemia (raised glucose levels) and subsequent decreased IRIS-1 expression may contribute to relative insulin deficiency in T2D patients. Similar isoforms were also identified in the mouse CD59B gene, and targeted CRISPR/Cas9-mediated knockout showed that these intracellular isoforms, but not canonical CD59B, are involved in insulin secretion from mouse β-cells. Mouse IRIS-2 is also down-regulated in diabetic db/db mouse islets. These findings establish the endogenous existence of previously undescribed non–GPI-anchored intracellular isoforms of human CD59 and mouse CD59B, which are required for normal insulin secretion.
Keywords: CD59; SNAREs; insulin secretion; intracellular complement; type 2 diabetes.
Conflict of interest statement
The authors declare no competing interest.
Figures


References
-
- Gonzalez L. L., Garrie K., Turner M. D., Type 2 diabetes: An autoinflammatory disease driven by metabolic stress. Biochim. Biophys. Acta Mol. Basis Dis. 1864, 3805–3823 (2018). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

