Clinical Implication of the Effect of the Production of Neutralizing Antibodies Against SARS-Cov-2 for Chronic Immune Thrombocytopenia Flare-Up Associated with COVID-19 Infection: A Case Report and the Review of Literature
- PMID: 35668857
- PMCID: PMC9166912
- DOI: 10.2147/IDR.S360238
Clinical Implication of the Effect of the Production of Neutralizing Antibodies Against SARS-Cov-2 for Chronic Immune Thrombocytopenia Flare-Up Associated with COVID-19 Infection: A Case Report and the Review of Literature
Abstract
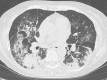
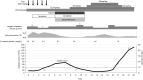
Previous studies have demonstrated that the appropriate production of serum anti-severe acute respiratory syndrome coronavirus 2 (SARS-Cov-2) neutralizing antibody (nAb) plays a critical role in the recovery from coronavirus disease 2019 (COVID-19); however, the role of nAb production in the recovery from a flare-up of chronic immune thrombocytopenia (ITP) has been unknown. We here report the first retrospectively investigated case of serum anti-SARS-Cov-2 nAb production during chronic ITP flare-up triggered by COVID-19. A 79-year-old woman with a history of corticosteroid-refractory ITP visited our hospital complaining of fever, cough, and sore throat for 4 days. Although chronic ITP was controlled by 12.5 mg of eltrombopag (EPAG) every other day, laboratory tests showed a decreased peripheral blood platelet count of 15.0 × 109/L, which indicated worsening thrombocytopenia. Meanwhile, PCR testing of a nasopharyngeal swab revealed that the patient was positive for SARS-Cov-2, and a computed tomography scan revealed bilateral pneumonia. On the basis of the flare-up of chronic ITP associated with COVID-19 pneumonia which was determined as a moderately severe status according to the WHO clinical progression scale, intravenous immunoglobulin therapy for 5 days (days 0-4) and antiviral therapy were added on top of EPAG, which only resulted in a transient increase in the platelet count for several days. After decreasing to 8.0 × 109/L on day 13, the platelet count increased from day 16, coinciding with a positive detection for serum nAb against SARS-Cov-2. Although the increased dose up to 50 mg/day of EPAG was challenged during the clinical course, rapid dose reduction did not cause another relapse. In addition, no thrombotic or bleeding event was seen. These collectively suggest the vital role of the production of anti-SARS-Cov-2 nAb and improvement of clinical symptoms for recovery from a flare-up of chronic ITP in our case.
Keywords: COVID-19; chronic immune thrombocytopenia; flare; neutralizing antibody.
© 2022 Maekura et al.
Conflict of interest statement
Taku Tsukamoto received research funding from Nippon Shinyaku. Tohru Inaba reports non-financial support from YHLO for technical support, during the conduct of the study. Junya Kuroda received research funding from Kyowa Kirin, Chugai Pharmaceutical, Ono Pharmaceutical, Sanofi, Eisai, Bristol-Myers Squibb (BMS), Sysmex, Dainippon Sumitomo Pharma, Nippon Shinyaku, AbbVie, Teijin, and Otsuka Pharmaceutical; received honoraria from Janssen Pharmaceutical K.K., Kyowa Kirin, Chugai Pharmaceutical, Ono Pharmaceutical, Sanofi, Eisai, Symbio, BMS, Astellas Pharma, Pfizer, Nippon Shinyaku, Daiichi Sankyo, Dainippon Sumitomo Pharma, AbbVie, and Otsuka Pharmaceutical; and is a consultant for Janssen Pharmaceutical K.K., and BMS. Other authors report no conflicts of interest in this work.
Figures
Similar articles
-
Treatment with eltrombopag of severe immune thrombocytopenia and hemolytic anemia associated with COVID-19 pneumonia: a case report.Ther Adv Hematol. 2021 Apr 27;12:20406207211011353. doi: 10.1177/20406207211011353. eCollection 2021. Ther Adv Hematol. 2021. PMID: 33995989 Free PMC article.
-
[Shedding of SARS-CoV-2 Virus in COVID-19 Patients and Neutralizing Antibody Level].Mikrobiyol Bul. 2022 Jul;56(3):416-431. doi: 10.5578/mb.20229704. Mikrobiyol Bul. 2022. PMID: 35960235 Turkish.
-
The SARS-CoV-2 Ivermectin Navarra-ISGlobal Trial (SAINT) to Evaluate the Potential of Ivermectin to Reduce COVID-19 Transmission in low risk, non-severe COVID-19 patients in the first 48 hours after symptoms onset: A structured summary of a study protocol for a randomized control pilot trial.Trials. 2020 Jun 8;21(1):498. doi: 10.1186/s13063-020-04421-z. Trials. 2020. PMID: 32513289 Free PMC article.
-
The role of anti-platelet factor 4 antibodies and platelet activation tests in patients with vaccine-induced immune thrombotic thrombocytopenia: Brief report on a comparison of the laboratory diagnosis and literature review.Clin Chim Acta. 2022 Apr 1;529:42-45. doi: 10.1016/j.cca.2022.02.003. Epub 2022 Feb 12. Clin Chim Acta. 2022. PMID: 35167842 Free PMC article. Review.
-
Immune thrombocytopenia and COVID-19: Case report and review of literature.Lupus. 2021 Aug;30(9):1515-1521. doi: 10.1177/09612033211021161. Epub 2021 May 30. Lupus. 2021. PMID: 34053365 Review.
Cited by
-
Successful Systemic Lysis Therapy of a Floating Carotid Thrombus in an Acute Stroke Patient with Known Immune Thrombocytopenia (ITP) on Ongoing Eltrombopag Therapy and Acute COVID-19 Infection: a Case Report.SN Compr Clin Med. 2023;5(1):133. doi: 10.1007/s42399-023-01465-5. Epub 2023 Apr 28. SN Compr Clin Med. 2023. PMID: 37151759 Free PMC article.
References
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

