Mecheliolide elicits ROS-mediated ERS driven immunogenic cell death in hepatocellular carcinoma
- PMID: 35671636
- PMCID: PMC9168183
- DOI: 10.1016/j.redox.2022.102351
Mecheliolide elicits ROS-mediated ERS driven immunogenic cell death in hepatocellular carcinoma
Abstract
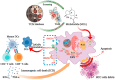
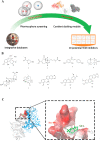
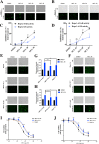
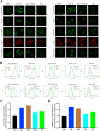
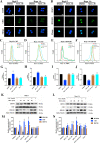
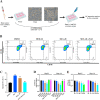
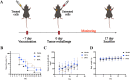
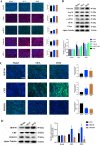
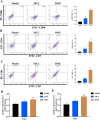
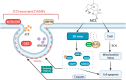
The nonnegligible reason for the poor prognosis of hepatocellular carcinoma (HCC) is resistance to conventional chemotherapy. Immunogenic cell death (ICD) is a rare immunostimulatory form of cell death that can reengage the tumor-specific immune system. ICD can improve the clinical outcomes of chemotherapeutics by promoting a long-term cancer immunity. The discovery of potential ICD inducers is emerging as a promising direction. In the present study, micheliolide (MCL), a natural guaianolide sesquiterpene lactone, was screened out by the virtual screening strategies, identified as an inhibitor of thioredoxin reductase (TrxR) and was evaluated to have high potential to induce ICD. Here, we showed that MCL induced ICD-associated DAMPs (damage-associated molecular patterns, such as CRT exposure, ATP secretion and HMGB1 release). MCL significantly triggered the regression of established tumors in an immunocompetent mouse vaccine model, and induced ICD (DCs maturation, the stimulation of CD4+, and CD8+ T-cells responses) in vivo. Mechanistically, we found that the magnitude of ICD-associated effects induced upon exposure of HCC cells to MCL was dependent on the generation of reactive oxygen species (ROS)-mediated endoplasmic reticulum stress (ERS). In addition, the suppression of ROS normalized MCL-induced ERS, in contrast, the downregulation of TrxR synergized with the ERS driven by MCL. We also systematically detected the H2O2 generation using Hyper7 sensors in HCC cells exposed to MCL. Notably, MCL inhibited the development of HCC organoids. Collectively, our results reveal a potential association between the TrxR inhibitors and ICD, presenting valuable insights into the MCL-activated ICD in HCC cells.
Copyright © 2022 The Authors. Published by Elsevier B.V. All rights reserved.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures









References
-
- Caiado F., Maia-Silva D., Jardim C., Schmolka N., Carvalho T., Reforco C., Faria R., Kolundzija B., Simões A.E., Baubec T., Vakoc C.R., da Silva M.G., Manz M.G., Schumacher T.N., Norell H., Silva-Santos B. Lineage tracing of acute myeloid leukemia reveals the impact of hypomethylating agents on chemoresistance selection. Nat. Commun. 2019;10:4986. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

