Synthesis and Glycosidation of Anomeric Halides: Evolution from Early Studies to Modern Methods of the 21st Century
- PMID: 35675037
- PMCID: PMC9417321
- DOI: 10.1021/acs.chemrev.2c00029
Synthesis and Glycosidation of Anomeric Halides: Evolution from Early Studies to Modern Methods of the 21st Century
Abstract
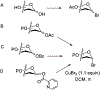
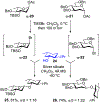
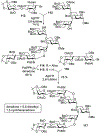
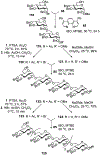
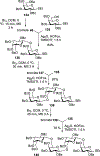
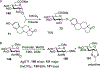
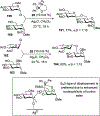
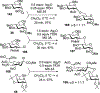
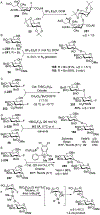
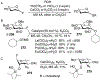
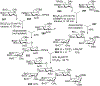
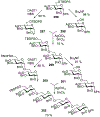
Advances in synthetic carbohydrate chemistry have dramatically improved access to common glycans. However, many novel methods still fail to adequately address challenges associated with chemical glycosylation and glycan synthesis. Since a challenge of glycosylation has remained, scientists have been frequently returning to the traditional glycosyl donors. This review is dedicated to glycosyl halides that have played crucial roles in shaping the field of glycosciences and continue to pave the way toward our understanding of chemical glycosylation.
Conflict of interest statement
The authors declare no competing financial interest.
Figures
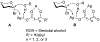
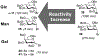

















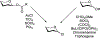








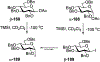













Similar articles
-
A brief account of the application of glycosyl halide as glycosyl radical precursor towards glycosylation through visible light catalysis.Carbohydr Res. 2025 Aug;554:109537. doi: 10.1016/j.carres.2025.109537. Epub 2025 May 17. Carbohydr Res. 2025. PMID: 40409016 Review.
-
Venturing beyond Donor-Controlled Glycosylation: New Perspectives toward Anomeric Selectivity.Acc Chem Res. 2018 Mar 20;51(3):628-639. doi: 10.1021/acs.accounts.7b00449. Epub 2018 Feb 22. Acc Chem Res. 2018. PMID: 29469568 Review.
-
Donor Preactivation-Based Glycan Assembly: from Manual to Automated Synthesis.Acc Chem Res. 2024 Jun 4;57(11):1577-1594. doi: 10.1021/acs.accounts.4c00072. Epub 2024 Apr 15. Acc Chem Res. 2024. PMID: 38623919
-
En Route to the Transformation of Glycoscience: A Chemist's Perspective on Internal and External Crossroads in Glycochemistry.J Am Chem Soc. 2021 Jan 13;143(1):17-34. doi: 10.1021/jacs.0c11106. Epub 2020 Dec 22. J Am Chem Soc. 2021. PMID: 33350830 Free PMC article.
-
Taming the Reactivity of Glycosyl Iodides To Achieve Stereoselective Glycosidation.Acc Chem Res. 2016 Jan 19;49(1):35-47. doi: 10.1021/acs.accounts.5b00357. Epub 2015 Nov 2. Acc Chem Res. 2016. PMID: 26524481
Cited by
-
Immunological and Toxicological Assessment of Triterpenoid Saponins Bearing Lewis-X- and QS-21-Based Trisaccharides.Chemistry. 2025 May 19;31(28):e202500994. doi: 10.1002/chem.202500994. Epub 2025 Apr 21. Chemistry. 2025. PMID: 40192644 Free PMC article.
-
Development of ketalized unsaturated saccharides as multifunctional cysteine-targeting covalent warheads.Commun Chem. 2024 Sep 9;7(1):201. doi: 10.1038/s42004-024-01279-z. Commun Chem. 2024. PMID: 39251816 Free PMC article.
-
Photosensitizer-free visible-light-promoted glycosylation enabled by 2-glycosyloxy tropone donors.Nat Commun. 2023 Dec 4;14(1):8025. doi: 10.1038/s41467-023-43786-y. Nat Commun. 2023. PMID: 38049421 Free PMC article.
-
Phenanthroline-Assisted Stereoselective Synthesis of 2-Deoxy Glycosides.ACS Omega. 2025 Apr 29;10(18):18700-18708. doi: 10.1021/acsomega.5c00189. eCollection 2025 May 13. ACS Omega. 2025. PMID: 40385149 Free PMC article.
-
Stereoselective glycosylation reactions with 2-deoxyglucose: a computational study of some catalysts.Comput Theor Chem. 2023 Jun;1224:114122. doi: 10.1016/j.comptc.2023.114122. Epub 2023 Apr 6. Comput Theor Chem. 2023. PMID: 37214423 Free PMC article.
References
-
- Hehre EJ Glycosyl transfer: a history of the concept’s development and view of its major contributions to biochemistry. Carbohydr. Res 2001, 331, 347–368. - PubMed
-
- Michael A On the synthesis of helicin and phenolglucoside. Am. Chem. J 1879, 1, 305–312.
-
- Fischer E Über die glucoside der alkohole. Ber. Dtsch. Chem. Ges 1893, 26, 2400–2412.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

