Controlling synthetic membraneless organelles by a red-light-dependent singlet oxygen-generating protein
- PMID: 35680863
- PMCID: PMC9184582
- DOI: 10.1038/s41467-022-30933-0
Controlling synthetic membraneless organelles by a red-light-dependent singlet oxygen-generating protein
Abstract
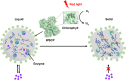
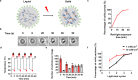
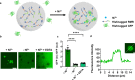
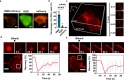
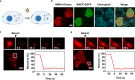
Membraneless organelles (MLOs) formed via protein phase separation have great implications for both physiological and pathological processes. However, the inability to precisely control the bioactivities of MLOs has hindered our understanding of their roles in biology, not to mention their translational applications. Here, by combining intrinsically disordered domains such as RGG and mussel-foot proteins, we create an in cellulo protein phase separation system, of which various biological activities can be introduced via metal-mediated protein immobilization and further controlled by the water-soluble chlorophyll protein (WSCP)-a remarkably stable, red-light-responsive singlet oxygen generator. The WSCP-laden protein condensates undergo a liquid-to-solid phase transition on light exposure, due to oxidative crosslinking, providing a means to control catalysis within synthetic MLOs. Moreover, these photoresponsive condensates, which retain the light-induced phase-transition behavior in living cells, exhibit marked membrane localization, reminiscent of the semi-membrane-bound compartments like postsynaptic densities in nervous systems. Together, this engineered system provides an approach toward controllable synthetic MLOs and, alongside its light-induced phase transition, may well serve to emulate and explore the aging process at the subcellular or even molecular level.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures




References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

