Autophagy Protects against Eosinophil Cytolysis and Release of DNA
- PMID: 35681515
- PMCID: PMC9180302
- DOI: 10.3390/cells11111821
Autophagy Protects against Eosinophil Cytolysis and Release of DNA
Abstract
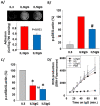
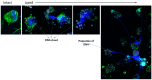
The presence of eosinophils in the airway is associated with asthma severity and risk of exacerbations. Eosinophils deposit their damaging products in airway tissue, likely by degranulation and cytolysis. We previously showed that priming blood eosinophils with IL3 strongly increased their cytolysis on aggregated IgG. Conversely, IL5 priming did not result in significant eosinophil cytolysis in the same condition. Therefore, to identify critical events protecting eosinophils from cell cytolysis, we examined the differential intracellular events between IL5- and IL3-primed eosinophils interacting with IgG. We showed that both IL3 and IL5 priming increased the eosinophil adhesion to IgG, phosphorylation of p38, and production of reactive oxygen species (ROS), and decreased the phosphorylation of cofilin. However, autophagic flux as measured by the quantification of SQSTM1-p62 and lipidated-MAP1L3CB over time on IgG, with or without bafilomycin-A1, was higher in IL5-primed compared to IL3-primed eosinophils. In addition, treatment with bafilomycin-A1, an inhibitor of granule acidification and autophagolysosome formation, enhanced eosinophil cytolysis and DNA trap formation in IL5-primed eosinophils. Therefore, this study suggests that increased autophagy in eosinophils protects from cytolysis and the release of DNA, and thus limits the discharge of damaging intracellular eosinophilic contents.
Keywords: DNA traps; IL3; IL5; IgG; MAP1LC3B; SQSTM1; autophagy; cytolysis; eosinophils.
Conflict of interest statement
Nizar N. Jarjour has received consulting fees from Glaxo Smith Kline (GSK), Astra-Zeneca, and Boehringer Ingelheim over the past three years. Sameer K. Mathur has received consulting and speaker fees from GSK and Astra-Zeneca over the past three years. These relationships with pharmaceutical companies are not relevant to the current study. The remaining authors declare no conflict of interest.
Figures




References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

