Exercise and Prebiotic Fiber Provide Gut Microbiota-Driven Benefit in a Survivor to Germ-Free Mouse Translational Model of Breast Cancer
- PMID: 35681702
- PMCID: PMC9179252
- DOI: 10.3390/cancers14112722
Exercise and Prebiotic Fiber Provide Gut Microbiota-Driven Benefit in a Survivor to Germ-Free Mouse Translational Model of Breast Cancer
Abstract
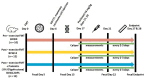
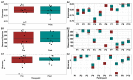
The gut microbiota plays a role in shaping overall host health and response to several cancer treatments. Factors, such as diet, exercise, and chemotherapy, can alter the gut microbiota. In the present study, the Alberta Cancer Exercise (ACE) program was investigated as a strategy to favorably modify the gut microbiota of breast cancer survivors who had received chemotherapy. Subsequently, the ability of post-exercise gut microbiota, alone or with prebiotic fiber supplementation, to influence breast cancer outcomes was interrogated using fecal microbiota transplant (FMT) in germ-free mice. While cancer survivors experienced little gut microbial change following ACE, in the mice, tumor volume trended consistently lower over time in mice colonized with post-exercise compared to pre-exercise microbiota with significant differences on days 16 and 22. Beta diversity analysis revealed that EO771 breast tumor cell injection and Paclitaxel chemotherapy altered the gut microbial communities in mice. Enrichment of potentially protective microbes was found in post-exercise microbiota groups. Tumors of mice colonized with post-exercise microbiota exhibited more favorable cytokine profiles, including decreased vascular endothelial growth factor (VEGF) levels. Beneficial microbial and molecular outcomes were augmented with prebiotic supplementation. Exercise and prebiotic fiber demonstrated adjuvant action, potentially via an enhanced anti-tumor immune response modulated by advantageous gut microbial shifts.
Keywords: breast cancer; chemotherapy; exercise; fecal microbiota transplant; gut microbiota; prebiotics.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures









References
-
- Scott A.J., Alexander J.L., Merrifield C.A., Cunningham D., Jobin C., Brown R., Alverdy J., Keefe S.J.O., Gaskins H.R., Teare J., et al. International Cancer Microbiome Consortium consensus statement on the role of the human microbiome in carcinogenesis. Gut. 2019;68:1624–1632. doi: 10.1136/gutjnl-2019-318556. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

