Colistin Monotherapy versus Colistin plus Meropenem Combination Therapy for the Treatment of Multidrug-Resistant Acinetobacter baumannii Infection: A Meta-Analysis
- PMID: 35683622
- PMCID: PMC9181109
- DOI: 10.3390/jcm11113239
Colistin Monotherapy versus Colistin plus Meropenem Combination Therapy for the Treatment of Multidrug-Resistant Acinetobacter baumannii Infection: A Meta-Analysis
Abstract
(1) Introduction: Colistin combination therapy with other antibiotics is a way to enhance colistin activity. The purpose of this meta-analysis was to compare the efficacy and safety of treatment with colistin monotherapy versus colistin plus meropenem combination therapy in patients with drug-resistant Acinetobacter baumannii infection. (2) Methods: All studies were included if they reported one or more of the following outcomes: clinical improvement, complete microbiological response, 14-day mortality, hospital mortality, or nephrotoxicity. (3) Results: Three randomized controlled trials and seven retrospective studies were included in the meta-analysis. Colistin monotherapy has similar rates of clinical improvement, 14-day mortality, hospital mortality, and nephrotoxicity as colistin plus meropenem combination therapy. Regarding complete microbiological response, the colistin plus meropenem combination was better than colistin monotherapy. (4) Discussion: Previous meta-analyses demonstrated heterogeneity in study quality and a lack of evidence supporting the use of colistin-based combination therapy. Our meta-analysis clearly showed that colistin combined with meropenem was not superior to colistin monotherapy for the treatment of Acinetobacter baumannii infection. (5) Conclusions: The efficacy and safety of treatment with colistin monotherapy and that of colistin plus meropenem combination therapy in patients with drug-resistant Acinetobacter baumannii infection were comparable. The majority of the evidence was obtained from nonrandomized studies, and high-quality randomized controlled trials are needed to confirm the role of colistin plus meropenem combination therapy in the treatment of multidrug-resistant Acinetobacter baumannii infection.
Keywords: Acinetobacter baumannii; colistin; combination therapy; meropenem; multidrug-resistant.
Conflict of interest statement
The authors declare that they have no competing interest.
Figures
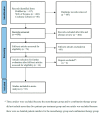



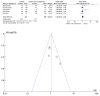


Similar articles
-
The effectiveness and nephrotoxicity of loading dose colistin combined with or without meropenem for the treatment of carbapenem-resistant A. baumannii.Int J Infect Dis. 2020 Aug;97:391-395. doi: 10.1016/j.ijid.2020.05.100. Epub 2020 Jun 2. Int J Infect Dis. 2020. PMID: 32502665
-
Comment on Huang et al. Colistin Monotherapy versus Colistin plus Meropenem Combination Therapy for the Treatment of Multidrug-Resistant Acinetobacter baumannii Infection: A Meta-Analysis. J. Clin. Med. 2022, 11, 3239.J Clin Med. 2022 Nov 28;11(23):7029. doi: 10.3390/jcm11237029. J Clin Med. 2022. PMID: 36498604 Free PMC article.
-
A Comparison of Colistin versus Colistin Plus Meropenem for the Treatment of Carbapenem-Resistant Acinetobacter baumannii in Critically Ill Patients: A Propensity Score-Matched Analysis.Antibiotics (Basel). 2020 Sep 28;9(10):647. doi: 10.3390/antibiotics9100647. Antibiotics (Basel). 2020. PMID: 32998187 Free PMC article.
-
Safety and efficacy of colistin alone or in combination in adults with Acinetobacter baumannii infection: A systematic review and meta-analysis.Int J Antimicrob Agents. 2019 Apr;53(4):383-400. doi: 10.1016/j.ijantimicag.2018.10.020. Epub 2018 Nov 14. Int J Antimicrob Agents. 2019. PMID: 30447379
-
Rifampicin as adjunct to colistin therapy in the treatment of multidrug-resistant Acinetobacter baumannii.Ann Pharmacother. 2014 Jun;48(6):766-71. doi: 10.1177/1060028014528306. Epub 2014 Mar 20. Ann Pharmacother. 2014. PMID: 24651164 Review.
Cited by
-
Flash optimization of drug combinations for Acinetobacter baumannii with IDentif.AI-AMR.NPJ Antimicrob Resist. 2025 Feb 21;3(1):12. doi: 10.1038/s44259-025-00079-2. NPJ Antimicrob Resist. 2025. PMID: 39984645 Free PMC article.
-
Treatment Strategies of Colistin Resistance Acinetobacter baumannii Infections.Antibiotics (Basel). 2024 May 6;13(5):423. doi: 10.3390/antibiotics13050423. Antibiotics (Basel). 2024. PMID: 38786151 Free PMC article. Review.
-
Gram Negatives and Antimicrobial Resistance: Two Faces of the Same Coin.J Clin Med. 2022 Sep 22;11(19):5574. doi: 10.3390/jcm11195574. J Clin Med. 2022. PMID: 36233444 Free PMC article.
-
Tackling Acinetobacter baumannii.J Clin Med. 2023 Aug 8;12(16):5168. doi: 10.3390/jcm12165168. J Clin Med. 2023. PMID: 37629210 Free PMC article.
-
Comparative phenotypic and proteomic analysis of colistin-exposed Pseudomonas aeruginosa.Germs. 2024 Sep 30;14(3):246-266. doi: 10.18683/germs.2024.1436. eCollection 2024 Sep. Germs. 2024. PMID: 39776958 Free PMC article.
References
Publication types
LinkOut - more resources
Full Text Sources

