Targeting TSLP in Asthma
- PMID: 35685846
- PMCID: PMC9172920
- DOI: 10.2147/JAA.S275039
Targeting TSLP in Asthma
Abstract
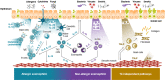
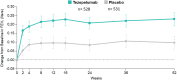
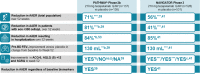
Thymic stromal lymphopoietin (TSLP) is an epithelial cell-derived cytokine implicated in the initiation and persistence of inflammatory pathways in asthma. Released in response to a range of epithelial insults (eg, allergens, viruses, bacteria, pollutants, and smoke), TSLP initiates multiple downstream innate and adaptive immune responses involved in asthma inflammation. Inhibition of TSLP is postulated to represent a novel approach to treating the diverse phenotypes and endotypes of asthma. Tezepelumab, the TSLP inhibitor farthest along in clinical development, is a human monoclonal antibody (IgG2λ) that binds specifically to TSLP, preventing interactions with its heterodimeric receptor. Results of recently published phase 2 and 3 studies, reviewed in this article, provide evidence of the safety and efficacy of tezepelumab that builds on initial findings. Tezepelumab is safe, well tolerated, and provides clinically meaningful improvements in asthma control, including reduced incidence of exacerbations and hospitalizations in patients with severe asthma. Clinical benefits were associated with reductions in levels of a broad spectrum of cytokines (eg, interleukin [IL]-5, IL-13) and baseline biomarkers (eg, blood eosinophils, immunoglobulin [Ig]E, fractional exhaled nitric oxide [FeNO]) and were observed across a range of severe asthma phenotypes (ie, eosinophilic and non-eosinophilic). These data strengthen the notion that anti-TSLP elicits broad inhibitory effects on pathways that are key to asthma inflammation rather than on narrower inhibition of individual downstream factors. This review presents the rationale for targeting TSLP to treat asthma, as well as the clinical effects of TSLP blockade on asthma outcomes, biomarkers of disease activity, airway inflammation, lung physiology, and patient symptoms.
Keywords: TSLP; anti-TSLP; asthma; exacerbation rates; thymic stromal lymphopoietin.
© 2022 Parnes et al.
Conflict of interest statement
Jane R Parnes reports a patent Treatment of asthma with anti-TSLP antibody: US-10828365-B2 issued to Assigned to Amgen and AstraZeneca. Jane R. Parnes and Nestor A. Molfino are employees and stockholders of Amgen Inc. Gene Colice and Ubaldo Martin are employees and stockholders of AstraZeneca. Jonathan Corren has received grant support, consulting fees, fees for a speakers bureau, and advisory board fees from AstraZeneca and Regeneron; grant support, advisory board fees, and fees for a speakers bureau from Genentech; and grant support from Sanofi, Teva Pharmaceutical Industries, and OptiNose. Andrew Menzies-Gow has received grants, advisory board fees, lecture fees, and consulting fees from AstraZeneca; advisory board fees from GlaxoSmithKline; advisory board fees and lecture fees from Novartis; advisory board fees, lecture fees, and travel expenses from Teva; advisory board fees, lecture fees, and consulting fees from Sanofi. The authors report no other conflicts of interest in this work.
Figures


References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

