Discovery of Tick-Borne Karshi Virus Implies Misinterpretation of the Tick-Borne Encephalitis Virus Seroprevalence in Northwest China
- PMID: 35685931
- PMCID: PMC9173002
- DOI: 10.3389/fmicb.2022.872067
Discovery of Tick-Borne Karshi Virus Implies Misinterpretation of the Tick-Borne Encephalitis Virus Seroprevalence in Northwest China
Abstract
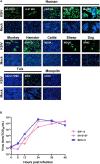
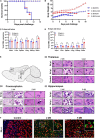
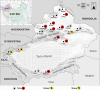
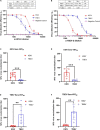
Despite few human cases of tick-borne encephalitis virus (TBEV), high rates of TBEV seroprevalence were reported among humans and animals in Xinjiang Uygur Autonomous Region in Northwestern China. In this study, the Karshi virus (KSIV) was identified and isolated from Hyalomma asiaticum ticks in Xinjiang. It belongs to the genus Flavivirus of the family Flaviviridae and is closely related to TBEV. KSIV infects cell lines from humans, other mammals and ticks, and causes encephalitis in suckling mice. High minimum infection rates (4.96%) with KSIV were detected among tick groups. KSIV infections have occurred in sheep and marmots, resulting in antibody-positive rates of 2.43 and 2.56%, respectively. We further found that, of the KSIV antibody-positive serum samples from animals, 13.9% had TBEV exposure showing cross-reaction to KSIV, and 11.1% had KSIV infection resulting in cross-reaction to TBEV; 8.3% were likely to have co-exposure to both viruses (or may be infected with one of them and present cross-reactivity with the other). The results revealed a substantial KSIV prevalence among ticks in Xinjiang, indicating exposure of animals to KSIV and TBEV. The findings implied misinterpretation of the high rates of TBEV seroprevalence among humans and animals in previous studies. There is a need to develop detection methods to distinguish KSIV from TBEV and to perform an in-depth investigation of KSIV and TBEV prevalence and incidence in Northwestern China, which would enhance our preparation to provide medical treatment of emerging diseases caused by tick-borne viral pathogens such as KSIV.
Keywords: Karshi virus; cross-reaction; prevalence; serological correlation; tick-borne encephalitis virus.
Copyright © 2022 Bai, Zhang, Su, Tang, Wang, Wu, Yang, Moming, Zhang, Bell-Sakyi, Sun, Shen and Deng.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
The Prevalence of Tick-Borne Encephalitis Virus in the Ticks and Humans of China from 2000 to 2023: A Systematic Review and Meta-Analysis.Vet Sci. 2025 Feb 8;12(2):146. doi: 10.3390/vetsci12020146. Vet Sci. 2025. PMID: 40005906 Free PMC article. Review.
-
[Detection of the Siberian Tick-borne Encephalitis Virus in the Xinjiang Uygur Autonomous Region, northwestern China].Bing Du Xue Bao. 2016 Jan;32(1):26-31. Bing Du Xue Bao. 2016. PMID: 27295880 Chinese.
-
Tick-Borne Encephalitis Virus Prevalence in Sheep, Wild Boar and Ticks in Belgium.Viruses. 2022 Oct 26;14(11):2362. doi: 10.3390/v14112362. Viruses. 2022. PMID: 36366458 Free PMC article.
-
First detection of TBE virus in ticks and sero-reactivity in goats in a non-endemic region in the southern part of Switzerland (Canton of Ticino).Ticks Tick Borne Dis. 2019 Jun;10(4):868-874. doi: 10.1016/j.ttbdis.2019.04.006. Epub 2019 Apr 18. Ticks Tick Borne Dis. 2019. PMID: 31047827
-
Geographic distribution of Tick-borne encephalitis virus complex.J Vector Borne Dis. 2020 Jan-Mar;57(1):14-22. doi: 10.4103/0972-9062.308794. J Vector Borne Dis. 2020. PMID: 33818450 Review.
Cited by
-
The Prevalence of Tick-Borne Encephalitis Virus in the Ticks and Humans of China from 2000 to 2023: A Systematic Review and Meta-Analysis.Vet Sci. 2025 Feb 8;12(2):146. doi: 10.3390/vetsci12020146. Vet Sci. 2025. PMID: 40005906 Free PMC article. Review.
-
The Known and Unknown of Global Tick-Borne Viruses.Viruses. 2024 Nov 21;16(12):1807. doi: 10.3390/v16121807. Viruses. 2024. PMID: 39772118 Free PMC article. Review.
-
Risk of infection with arboviruses in a healthy population in Pakistan based on seroprevalence.Virol Sin. 2024 Jun;39(3):369-377. doi: 10.1016/j.virs.2024.04.001. Epub 2024 Apr 8. Virol Sin. 2024. PMID: 38599520 Free PMC article.
-
Diversity of species and geographic distribution of tick-borne viruses in China.Front Microbiol. 2024 Feb 27;15:1309698. doi: 10.3389/fmicb.2024.1309698. eCollection 2024. Front Microbiol. 2024. PMID: 38476950 Free PMC article.
References
-
- Apanaskevich D. A., Filippova N. A., Horak I. G. (2010). The genus Hyalomma Koch, 1844. x. redescription of all parasitic stages of H. (Euhyalomma) scupense Schulze, 1919 (=H. detritum Schulze) (Acari: Ixodidae) and notes on its biology. Folia Parasitol. 57 69–78. 10.14411/fp.2010.009 - DOI - PubMed
-
- Aristova V. A., Gushchina E. A., Gromashevskii V. L., Gushchin B. V. (1986). Experimental infection of ixodid ticks with Karshi virus. Parazitologiia 20 347–350. - PubMed
-
- Bi W., Deng H., Bu X. (1997). Regionalisation of natural foci of forest encephalitis. Shou Du Shi Fan Da Xue Xue Bao 18 100–107.
LinkOut - more resources
Full Text Sources

