The impact of cardiovascular comorbidities associated with risk for left heart disease on idiopathic pulmonary arterial hypertension: Data from the Hellenic Pulmonary Hypertension Registry (HOPE)
- PMID: 35685948
- PMCID: PMC9171835
- DOI: 10.1002/pul2.12086
The impact of cardiovascular comorbidities associated with risk for left heart disease on idiopathic pulmonary arterial hypertension: Data from the Hellenic Pulmonary Hypertension Registry (HOPE)
Abstract
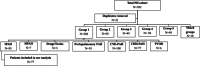
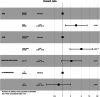
Whereas younger female patients were diagnosed with idiopathic pulmonary arterial hypertension (IPAH) in 1980s, it is now frequently encountered in elderly patients with cardiovascular comorbidities (CVCs) associated with increased risk for left heart disease. We present data until November 2019 regarding specific features and clinical outcomes of IPAH population from the Hellenic Pulmonary Hypertension Registry (HOPE). Patients were divided into two groups based on the presence of ≥ or <3 CVCs, arterial hypertension, diabetes mellitus, obesity, presence of coronary artery disease, or atrial fibrillation. Overall, 77 patients with IPAH (55.1 [interquartile range, IQR: 24.1] years, 62.8% women) have been recorded. Fifteen patients (19.2%) had ≥3 CVCs, while 25 (32%) were over 65 years old. Patients with ≥3 CVCs were older, presented an almost equal female to male ratio, walked less in 6-min walk test, and had lower mean arterial pulmonary pressure and pulmonary vascular resistance at baseline than patients with less CVCs. Fewer patients with ≥3 CVCs received PAH-specific treatment compared to patients with less comorbidities (n = 11 [73.3%] versus n = 58 [95.5%], p = 0.02). During a median follow-up period of 3.8 (IQR: 2.7) years, 18 patients died (all-cause mortality 24.3%). Male sex and older age were independent predictors of mortality and/or lung transplantation, while CVCs did not have a significant impact on clinical outcomes. In this nationwide, register-based study, the epidemiology of IPAH involves older patients with CVCs, who seem to have less hemodynamic compromise, but worse functional impairment and are treated less aggressively with PAH pharmacotherapy.
Keywords: cardiovascular comorbidities; elderly; idiopathic pulmonary arterial hypertension; survival; targeted therapy.
© 2022 The Authors. Pulmonary Circulation published by John Wiley & Sons Ltd on behalf of Pulmonary Vascular Research Institute.
Conflict of interest statement
Alexandra Arvanitaki has been the recipient of the International Training and Research Fellowship EMAH Stiftung Karla Voellm, Krefeld, Germany. Anastasia Anthi reports receiving honoraria and consultancy fees from Actelion Pharmaceuticals Hellas, Bayer, ELPEN, GSK, MSD, Lilly, and United Therapeutics. Eftychia Demerouti has been an advisory board member for Actelion Pharmaceuticals Hellas, MSD Hellas, and GlaxoSmithKline and an honorarium speaker for Actelion Pharmaceutical Hellas and MSD Hellas. Ioanna Mitrouska reports receiving honoraria and consultancy fees from Actelion Pharmaceuticals Hellas, Bayer, ELPEN, MSD, and GSK. Katerina K. Naka reports lecture fees from Novartis, Medtronic, and Abbott, while she serves as an investigator in randomized clinical trials and/or registries sponsored by Novartis, Merck, Amgen, BMS, Boehringer Ingelheim, Pfizer, and Actelion Pharmaceutical Hellas, outside the submitted work. Stylianos E. Orfanos reports has been receiving research grants and/or honoraria and/or consultancy fees from Actelion Pharmaceutical Hellas, Bayer, ELPEN, Galenica‐Ferrer, GSK, MSD, Pharmaserve Lilly, PharmaSwiss, Pfizer, and United Therapeutics. Georgia Pitsiou reports receiving honoraria and consultancy fees from Actelion Pharmaceutical Hellas, Bayer, ELPEN, MSD, and GSK. Ioannis Stanopoulos reports receiving honoraria and consultancy fees from Actelion Pharmaceuticals Hellas. Iraklis Tsangaris reports has been receiving honoraria and consultancy fees from Actelion Pharmaceuticals Hellas, Bayer, ELPEN, GSK, Pfizer, Lilly, and United Therapeutics. Athanasios Manginas reports consultation fees from Astra Zeneca, Bayer, ELPEN, Actelion Pharmaceuticals Hellas, and MSD. George Giannakoulas reports has been receiving honoraria and consultancy fees from Actelion Pharmaceuticals Hellas, Bayer, ELPEN, Galenica‐Ferrer, GSK, Pfizer, Lilly, MSD, and United Therapeutics. The remaining authors declare no conflicts of interest. The remaining authors declare no conflicts of interest.
Figures


References
-
- Galiè N, Humbert M, Vachiery JL, Gibbs S, Lang I, Torbicki A, Simonneau G, Peacock A, Vonk Noordegraaf A, Beghetti M, Ghofrani A, Gomez Sanchez MA, Hansmann G, Klepetko W, Lancellotti P, Matucci M, McDonagh T, Pierard LA, Trindade PT, Zompatori M, Hoeper M, ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension: The Joint Task Force for the Diagnosis and Treatment of Pulmonary Hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS): Endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC), International Society for Heart and Lung Transplantation (ISHLT). Eur Respir J. 2015;46:903–75. 10.1183/13993003.01032-2015 - DOI - PubMed
-
- Arvanitaki A, Boutsikou M, Anthi A, Apostolopoulou S, Avgeropoulou A, Demerouti E, Farmakis D, Feloukidis C, Giannakoulas G, Karvounis H, Karyofyllis P, Mitrouska I, Mouratoglou SA, Naka KK, Orfanos SE, Panagiotidou E, Pitsiou G, Rammos S, Stagaki E, Stanopoulos I, Thomaidi A, Triantafyllidi H, Tsangaris I, Tsiapras D, Voudris V, Manginas A, Hellenic Society for the Study of Pulmonary Hypertension . Epidemiology and initial management of pulmonary arterial hypertension: real‐world data from the Hellenic pulmOnary hyPertension rEgistry (HOPE). Pulm Circ. 2019;9:2045894019877157. 10.1177/2045894019877157 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources

