Three-way analysis-based pH-UV-Vis spectroscopy for quantifying allura red in an energy drink and determining colorant's pKa
- PMID: 35696222
- PMCID: PMC9261852
- DOI: 10.38212/2224-6614.1275
Three-way analysis-based pH-UV-Vis spectroscopy for quantifying allura red in an energy drink and determining colorant's pKa
Abstract
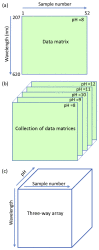
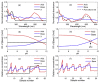
Three-way analysis-based pH-UV-Vis spectroscopy was proposed for quantifying allura red in an energy drink product without the need for chromatographic analysis, and determining the colorant's pKa without using any titration technique. In this study, UV-Vis spectroscopic data matrices were obtained from absorbance measurements at five different pH levels from pH 8 to pH 12 and arranged as a three-way array (wavelength × sample × pH). In the three-way analysis procedure, parallel factor analysis (PARAFAC) was implemented to decompose the three-way array into a set of trilinear components. Each set of three components relates to spectral, pH and relative concentration profiles of allura red and sample matrix in the energy drink. First, UV-Vis spectra of the colorant's acid-base pair and sample's matrix were characterized by using the estimated spectral profile. Then, from the pH profile the pKa value was found to be 11.28 for the related colorant. Finally, allura red in energy drink samples was determined using the estimated concentration curve in the relative concentration profile. In the quantitation procedure, the working concentration range was 0.8-19.2 μg/mL. PARAFAC approach was tested in terms of selectivity, precision, and accuracy of the method. Added recovery results obtained by applying the proposed method to spiked samples were between 101.5% and 103.5%. In the application of the method to the analysis of real samples, successful results were reported. For a comparison, an ultra-performance liquid chromatographic method was developed for the quantitation of the colorant. Compared to the chromatographic method, we observed that PARAFAC model was simple and less expensive without requiring separation.
Figures



References
-
- Riolo D, Piazza A, Cottini C, Serafini M, Lutero E, Cuoghi E, Gasparini L, Botturi D, Marino IG, Aliatis I, Bersani D, Lottici PP. Raman spectroscopy as a PAT for pharmaceutical blending: Advantages and disadvantages. J Pharmaceut Biomed. 2018;149:329–34. - PubMed
-
- Li Y, Shen Y, Yao C-l, Guo D-a. Quality assessment of herbal medicines based on chemical fingerprints combined with chemometrics approach: A review. J Pharmaceut Biomed. 2020;185:113215. - PubMed
-
- Power AC, Chapman J, Chandra S, Cozzolino D. Ultraviolet-visible spectroscopy for food quality analysis. Eval Tech Food Qual. 2019:91–104.
-
- Arancibia JA, Damiani PC, Escandar GM, Ibañez GA, Olivieri AC. A review on second- and third-order multivariate calibration applied to chromatographic data. J Chromatogr B. 2012;910:22–30. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
