Effect of Varenicline Added to Counseling on Smoking Cessation Among African American Daily Smokers: The Kick It at Swope IV Randomized Clinical Trial
- PMID: 35699705
- PMCID: PMC9198729
- DOI: 10.1001/jama.2022.8274
Effect of Varenicline Added to Counseling on Smoking Cessation Among African American Daily Smokers: The Kick It at Swope IV Randomized Clinical Trial
Abstract
Importance: African American smokers have among the highest rates of tobacco-attributable morbidity and mortality in the US, and effective treatment is needed for all smoking levels.
Objectives: To evaluate the efficacy of varenicline vs placebo among African American adults who are light, moderate, and heavy daily smokers.
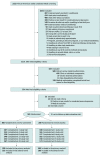
Design, setting, and participants: The Kick It at Swope IV (KIS-IV) trial was a randomized, double-blind, placebo-controlled clinical trial conducted at a federally qualified health center in Kansas City. A total of 500 African American adults who were daily smokers of all smoking levels were enrolled from June 2015 to December 2017; final follow-up was completed in June 2018.
Interventions: Participants were provided 6 sessions of culturally relevant individualized counseling and were randomized (in a 3:2 ratio) to receive varenicline (1 mg twice daily; n = 300) or placebo (n = 200) for 12 weeks. Randomization was stratified by sex and smoking level (1-10 cigarettes/d [light smokers] or >10 cigarettes/d [moderate to heavy smokers]).
Main outcomes and measures: The primary outcome was salivary cotinine-verified 7-day point prevalence smoking abstinence at week 26. The secondary outcome was 7-day point prevalence smoking abstinence at week 12, with subgroup analyses for light smokers (1-10 cigarettes/d) and moderate to heavy smokers (>10 cigarettes/d).
Results: Among 500 participants who were randomized and completed the baseline visit (mean age, 52 years; 262 [52%] women; 260 [52%] light smokers; 429 [86%] menthol users), 441 (88%) completed the trial. Treating those lost to follow-up as smokers, participants receiving varenicline were significantly more likely than those receiving placebo to be abstinent at week 26 (15.7% vs 6.5%; difference, 9.2% [95% CI, 3.8%-14.5%]; odds ratio [OR], 2.7 [95% CI, 1.4-5.1]; P = .002). The varenicline group also demonstrated greater abstinence than the placebo group at the end of treatment week 12 (18.7% vs 7.0%; difference, 11.7% [95% CI, 6.0%-17.7%]; OR, 3.0 [95% CI, 1.7-5.6]; P < .001). Smoking abstinence at week 12 was significantly greater for individuals receiving varenicline compared with placebo among light smokers (22.1% vs 8.5%; difference, 13.6% [95% CI, 5.2%-22.0%]; OR, 3.0 [95% CI, 1.4-6.7]; P = .004) and among moderate to heavy smokers (15.1% vs 5.3%; difference, 9.8% [95% CI, 2.4%-17.2%]; OR, 3.1 [95% CI, 1.1-8.6]; P = .02), with no significant smoking level × treatment interaction (P = .96). Medication adverse events were generally comparable between treatment groups, with nausea reported more frequently in the varenicline group (163 of 293 [55.6%]) than the placebo group (90 of 196 [45.9%]).
Conclusions and relevance: Among African American adults who are daily smokers, varenicline added to counseling resulted in a statistically significant improvement in the rates of 7-day point prevalence smoking abstinence at week 26 compared with counseling and placebo. The findings support the use of varenicline in addition to counseling for tobacco use treatment among African American adults who are daily smokers.
Trial registration: ClinicalTrials.gov Identifier: NCT02360631.
Conflict of interest statement
Figures

Comment in
-
Helping African American Individuals Quit Smoking: Finally, Some Progress.JAMA. 2022 Jun 14;327(22):2192-2194. doi: 10.1001/jama.2022.9161. JAMA. 2022. PMID: 35699718 No abstract available.
References
-
- A Socioecological Approach to Addressing Tobacco-Related Health Disparities. National Cancer Institute; 2017.

