Unraveling the role of Epac1-SOCS3 signaling in the development of neonatal-CRD-induced visceral hypersensitivity in rats
- PMID: 35702948
- PMCID: PMC9344090
- DOI: 10.1111/cns.13880
Unraveling the role of Epac1-SOCS3 signaling in the development of neonatal-CRD-induced visceral hypersensitivity in rats
Abstract
Aims: Visceral hypersensitivity in irritable bowel syndrome (IBS) is widespread, but effective therapies for it remain elusive. As a canonical anti-inflammatory protein, suppressor of cytokine signaling 3 (SOCS3) reportedly relays exchange protein 1 directly activated by cAMP (Epac1) signaling and inhibits the intracellular response to inflammatory cytokines. Despite the inhibitory effect of SOCS3 on the pro-inflammatory response and neuroinflammation in PVN, the systematic investigation of Epac1-SOCS3 signaling involved in visceral hypersensitivity remains unknown. This study aimed to explore Epac1-SOCS3 signaling in the activity of hypothalamic paraventricular nucleus (PVN) corticotropin-releasing factor (CRF) neurons and visceral hypersensitivity in adult rats experiencing neonatal colorectal distension (CRD).
Methods: Rats were subjected to neonatal CRD to simulate visceral hypersensitivity to investigate the effect of Epac1-SOCS3 signaling on PVN CRF neurons. The expression and activity of Epac1 and SOCS3 in nociceptive hypersensitivity were determined by western blot, RT-PCR, immunofluorescence, radioimmunoassay, electrophysiology, and pharmacology.
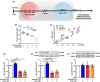
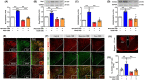
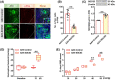
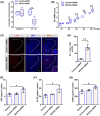
Results: In neonatal-CRD-induced visceral hypersensitivity model, Epac1 and SOCS3 expressions were downregulated and IL-6 levels elevated in PVN. However, infusion of Epac agonist 8-pCPT in PVN reduced CRF neuronal firing rates, and overexpression of SOCS3 in PVN by AAV-SOCS3 inhibited the activation of PVN neurons, reduced visceral hypersensitivity, and precluded pain precipitation. Intervention with IL-6 neutralizing antibody also alleviated the visceral hypersensitivity. In naïve rats, Epac antagonist ESI-09 in PVN increased CRF neuronal firing. Consistently, genetic knockdown of Epac1 or SOCS3 in PVN potentiated the firing rate of CRF neurons, functionality of HPA axis, and sensitivity of visceral nociception. Moreover, pharmacological intervention with exogenous IL-6 into PVN simulated the visceral hypersensitivity.
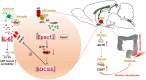
Conclusions: Inactivation of Epac1-SOCS3 pathway contributed to the neuroinflammation accompanied by the sensitization of CRF neurons in PVN, precipitating visceral hypersensitivity and pain in rats experiencing neonatal CRD.
Keywords: Epac1; SOCS3; corticotrophin-releasing factor; neonatal colorectal distension; visceral hypersensitivity.
© 2022 The Authors. CNS Neuroscience & Therapeutics published by John Wiley & Sons Ltd.
Conflict of interest statement
The authors declared no conflict of interests.
Figures


Similar articles
-
Activation of corticotropin-releasing factor neurons and microglia in paraventricular nucleus precipitates visceral hypersensitivity induced by colorectal distension in rats.Brain Behav Immun. 2016 Jul;55:93-104. doi: 10.1016/j.bbi.2015.12.022. Epub 2015 Dec 29. Brain Behav Immun. 2016. PMID: 26743854
-
Disinhibition of PVN-projecting GABAergic neurons in AV region in BNST participates in visceral hypersensitivity in rats.Psychoneuroendocrinology. 2020 Jul;117:104690. doi: 10.1016/j.psyneuen.2020.104690. Epub 2020 Apr 24. Psychoneuroendocrinology. 2020. PMID: 32417623
-
Mast cells in the paraventricular nucleus participate in visceral hypersensitivity induced by neonatal maternal separation.Behav Brain Res. 2021 Mar 26;402:113113. doi: 10.1016/j.bbr.2020.113113. Epub 2021 Jan 4. Behav Brain Res. 2021. PMID: 33412227
-
Corticotrophin-releasing factor 1 activation in the central amygdale and visceral hyperalgesia.Neurogastroenterol Motil. 2015 Jan;27(1):1-6. doi: 10.1111/nmo.12495. Neurogastroenterol Motil. 2015. PMID: 25557223 Free PMC article. Review.
-
Role of corticotropin-releasing factor pathways in stress-related alterations of colonic motor function and viscerosensibility in female rodents.Gend Med. 2005 Sep;2(3):146-54. doi: 10.1016/s1550-8579(05)80043-9. Gend Med. 2005. PMID: 16290887 Review.
Cited by
-
Role of EPAC1 in chronic pain.Biochem Biophys Rep. 2024 Jan 22;37:101645. doi: 10.1016/j.bbrep.2024.101645. eCollection 2024 Mar. Biochem Biophys Rep. 2024. PMID: 38304575 Free PMC article. Review.
-
Paraventricular thalamus-insular cortex circuit mediates colorectal visceral pain induced by neonatal colonic inflammation in mice.CNS Neurosci Ther. 2024 Apr;30(4):e14534. doi: 10.1111/cns.14534. Epub 2023 Nov 23. CNS Neurosci Ther. 2024. PMID: 37994678 Free PMC article.
-
Thalamic Nucleus Reuniens Glutamatergic Neurons Mediate Colorectal Visceral Pain in Mice via 5-HT2B Receptors.Neurosci Bull. 2024 Oct;40(10):1421-1433. doi: 10.1007/s12264-024-01207-0. Epub 2024 May 13. Neurosci Bull. 2024. PMID: 38739251 Free PMC article.
-
Electroacupuncture in the treatment of IBS in rats: investigation of the mechanisms of CRH+ neurons in the paraventricular nucleus.J Neurophysiol. 2023 Aug 1;130(2):380-391. doi: 10.1152/jn.00156.2023. Epub 2023 Jul 12. J Neurophysiol. 2023. PMID: 37435647 Free PMC article.
-
[Mechanisms by Which Paraventricular Hypothalamic Nucleus Participates in the Acupuncture Treatment of Diseases].Sichuan Da Xue Xue Bao Yi Xue Ban. 2025 Jan 20;56(1):26-34. doi: 10.12182/20250160202. Sichuan Da Xue Xue Bao Yi Xue Ban. 2025. PMID: 40109474 Free PMC article. Review. Chinese.
References
-
- Solovyova G, Kvacheniuk K, Vlasyuk S, Antonyuk O. Capsumen in the treatment of irritable bowel syndrome. Georgian Med News. 2019;289:94‐102. - PubMed
-
- Ludidi S, Conchillo JM, Keszthelyi D, et al. Rectal hypersensitivity as hallmark for irritable bowel syndrome: defining the optimal cutoff . Neurogastroenterol Motil. 2012;24(8):729‐733. e345‐6. - PubMed
-
- Greenwood‐van Meerveld B. Importance of 5‐hydroxytryptamine receptors on intestinal afferents in the regulation of visceral sensitivity . Neurogastroenterol Motil. 2007;19(Suppl 2):13‐18. - PubMed
-
- Barbara G, Cremon C, de Giorgio R, et al. Mechanisms underlying visceral hypersensitivity in irritable bowel syndrome . Curr Gastroenterol Rep. 2011;13(4):308‐315. - PubMed
-
- Anand P, Aziz Q, Willert R, van Oudenhove L. Peripheral and central mechanisms of visceral sensitization in man . Neurogastroenterol Motil. 2007;19(1 Suppl):29‐46. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

