Peripheral blood CD4posCD25posFoxP3pos cells and inflammatory cytokines as biomarkers of response in rheumatoid arthritis patients treated with CTLA4-Ig
- PMID: 35706043
- PMCID: PMC9199213
- DOI: 10.1186/s13075-022-02827-5
Peripheral blood CD4posCD25posFoxP3pos cells and inflammatory cytokines as biomarkers of response in rheumatoid arthritis patients treated with CTLA4-Ig
Erratum in
-
Correction: Peripheral blood CD4posCD25posFoxP3pos cells and inflammatory cytokines as biomarkers of response in rheumatoid arthritis patients treated with CTLA4-Ig.Arthritis Res Ther. 2022 Jul 25;24(1):177. doi: 10.1186/s13075-022-02866-y. Arthritis Res Ther. 2022. PMID: 35879726 Free PMC article. No abstract available.
Abstract
Background: Prognostic biomarkers of treatment response to distinct biologic disease-modifying anti-rheumatic drugs (b-DMARDs) are still lacking within the management of rheumatoid arthritis (RA).
Methods: Thirty-four b-DMARDs naive RA patients, divided by disease duration into early (cohort 1) and long standing (cohort 2), received CTLA4-Ig. At study entry, and every 3 months for 1 year, each patient underwent peripheral blood (PB)-derived CD4pos cell subpopulation assessment by flow cytometry, STAT3 and STAT5 expression by RT-PCR and IL-6, IL-12p70, TGFβ, and IL-10 serum levels by ELISA. The DAS and CDAI remission was assessed at 6 and 12 months.
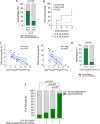
Results: DAS- and CDAI-defined remission within 12 months was achieved by 16 (47.1%) and 8 (23.5%) RA patients, respectively. Considering the whole RA cohort, CTLA4-Ig induced a significant decrease of IL-6 serum levels from baseline to 6 and 12 months, as well as of PB CD4posCD25posFoxP3pos cells at 6 and 12 months, and of CD4posIL17pos cells after 12 months. PB CD4pos cells of RA patients showed higher STAT3 and STAT5 expression than healthy controls, which remained unchanged within 12 months of treatment. At study entry, RA patients achieving DAS remission had significantly lower IL-6 serum levels than RA patients not achieving this outcome. In particular, having baseline IL-6 serum levels ≤ 8.4 pg/ml, significantly identified naïve to b-DMARDs RA patients more likely to achieve DAS-remission under CTLA4-Ig at 6 months (66.7%) compared to RA patients with baseline IL-6 serum levels > 8.4 pg/ml [15.4%, OR (95%Cis) 11.00 (1.75-55.82)]. Moreover, having CD4posCD25posFoxP3pos cells rate ≥ 6.0% significantly identifies naïve to b-DMARDs early RA patients more likely to achieve DAS remission at 6 months (83.3%) compared to RA patients with baseline CD4posCD25posFoxP3pos cells < 6.0% [16.7%, OR (95% Cis) 25.00 (1.00-336.81)].
Conclusions: Baseline IL-6 serum levels and peripheral blood-derived CD4pos subpopulations are putative novel prognostic biomarkers of CTLA4-Ig response in RA patients.
Keywords: Biomarkers; CTLA4-Ig; Rheumatoid arthritis.
© 2022. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures



References
-
- Kerschbaumer A, Sepriano A, Smolen JS, van der Heijde D, Dougados M, van Vollenhoven R, et al. Efficacy of pharmacological treatment in rheumatoid arthritis: a systematic literature research informing the 2019 update of the EULAR recommendations for management of rheumatoid arthritis. Ann Rheum Dis. 2020;79:744–759. doi: 10.1136/annrheumdis-2019-216656. - DOI - PMC - PubMed
-
- Humby F, Lewis M, Ramamoorthi N, Hackney JA, Barnes MR, Bombardieri M, et al. Synovial cellular and molecular signatures stratify clinical response to csDMARD therapy and predict radiographic progression in early rheumatoid arthritis patients. Ann Rheum Dis. 2019;78:761–772. doi: 10.1136/annrheumdis-2018-214539. - DOI - PMC - PubMed
-
- Alivernini S, Tolusso B, Gessi M, Gigante MR, Mannocci A, Petricca L, et al. Inclusion of synovial tissue-derived characteristics in a nomogram for the prediction of treatment response in treatment-naive rheumatoid arthritis patients. Arthritis Rheumatol. 2021;73:1601–1613. doi: 10.1002/art.41726. - DOI - PMC - PubMed
-
- Aldridge J, Pandya JM, Meurs L, Andersson K, Nordström I, Theander E, et al. Sex-based differences in association between circulating T cell subsets and disease activity in untreated early rheumatoid arthritis patients. Arthritis Res Ther. 2018;20:150. doi: 10.1186/s13075-018-1648-2. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

