The efficacy and safety of concurrent chemoradiotherapy with induction chemotherapy vs. concurrent chemoradiotherapy alone for locally advanced nasopharyngeal carcinoma: a systematic-review and meta-analysis
- PMID: 35706809
- PMCID: PMC9189232
- DOI: 10.21037/tcr-22-604
The efficacy and safety of concurrent chemoradiotherapy with induction chemotherapy vs. concurrent chemoradiotherapy alone for locally advanced nasopharyngeal carcinoma: a systematic-review and meta-analysis
Abstract
Background: Although nasopharyngeal carcinoma (NPC) is very sensitive to radiotherapy, patients with advanced NPC still need concurrent chemoradiotherapy (CCRT). The induction chemotherapy (IC) before CCRT can possiblly reduce the distant metastasis rate and improve the survival rate, but the results of studies varied. In this meta-analysis, we included controlled clinical studies to systematically evaluate the gain effect of it.
Methods: The PubMed, Web of Science, Wiley online library, Elsevier, CNKI, and Clinicaltrials.gov databases were electronically searched using keywords "neoadjuvant chemotherapy/induction chemotherapy" and "concurrent chemoradiotherapy" and "nasopharyngeal carcinoma" for articles from the date of establishment of the database to January, 2022. The inclusion criteria was established according to the PICOS principles. The complete response rate, progression-free survival (PFS), distant metastasis-free survival (DMFS), and overall survival (OS) were used as the main efficacy indicators for analysis.
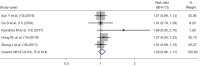
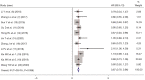
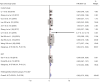
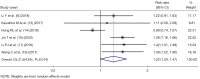
Results: Thirteen articles were included with a total of 7,197 patients, including 3,764 patients who took IC + CCRT and 3,433 patients who received CCRT alone. Two randomized controlled trials (RCTs) included had low risk of bias and 11 cohort studies had some concerns of risk. Meta-analysis showed that IC did not significantly increase the complete response rate [risk ratio (RR) =1.03, 95% confidence interval (CI): 0.96-1.11, P=0.336], the pooled hazard ratio (HR) of IC as a prognostic factor predicting PFS was (HR =0.87, 95% CI: 0.78-0.96, P=0.214), and DMFS (HR =0.76, 95% CI: 0.57-0.96, P=0.004), and OS (HR =0.82, 95% CI: 0.73-0.92, P=0.036). IC increased the adverse effects experienced by patients (RR =1.22, 95% CI: 1.00-1.47, P=0.045).
Discussion: The use of IC before CCRT provided a good effect for NPC patients with tumor, node & metastasis (TNM) stages III and IV, but there was no significant gain for patients with stage II. In addition, the application of IC will increase the toxicity in the oral cavity, digestive tract, and bone marrow, and should be well considered about the toleration of patients. Due to the limitations of this study, more literatures with better quality are needed for further explore into this topic.
Keywords: Local nasopharyngeal carcinoma; concurrent chemoradiotherapy (CCRT); induction chemotherapy.
2022 Translational Cancer Research. All rights reserved.
Conflict of interest statement
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://tcr.amegroups.com/article/view/10.21037/tcr-22-604/coif). The authors have no conflicts of interest to declare.
Figures




References
LinkOut - more resources
Full Text Sources
