Comprehensive Analysis Identifies PI3K/Akt Pathway Alternations as an Immune-Related Prognostic Biomarker in Colon Adenocarcinoma Patients Receiving Immune Checkpoint Inhibitor Treatment
- PMID: 35707003
- PMCID: PMC9192307
- DOI: 10.1155/2022/8179799
Comprehensive Analysis Identifies PI3K/Akt Pathway Alternations as an Immune-Related Prognostic Biomarker in Colon Adenocarcinoma Patients Receiving Immune Checkpoint Inhibitor Treatment
Abstract
Introduction: In recent years, immune checkpoint inhibitors (ICIs) have attracted widespread attention and made breakthroughs in progress towards the treatment of various cancers. However, ICI therapy is selective, and its effects on many patients are not ideal. It is therefore critical to identify prognostic biomarkers of response to ICI therapy. The PI3K/Akt pathway plays important roles in tumor formation and metastasis. However, there are no published reports clarifying the relationship between PI3K/Akt pathway mutations and prognosis for colon adenocarcinoma (COAD) patients receiving immunotherapy.
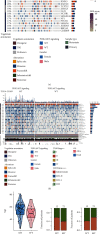
Methods: We collected data from a COAD cohort from The Cancer Genome Atlas (TCGA) database, including whole-exome sequencing (WES) data, RNA-seq data, and clinical data. We also collected data, including clinical prognosis and targeted sequencing data, from a cohort of COAD patients receiving immunotherapy. We collected 50 COAD patients (Local-COAD) from the Zhujiang Hospital of Southern Medical University and performed targeted sequencing. We analyzed the effects of PI3K/Akt pathway mutations on the patients' clinical prognosis, immunogenicity, and immune microenvironments. Gene set enrichment analysis (GSEA) was used to analyze the significantly upregulated and downregulated signaling pathways. We used these results to hypothesize potential mechanisms by which PI3K/Akt mutations could affect the prognosis of COAD patients.
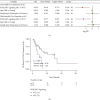
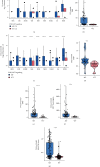
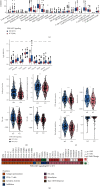
Results: Univariate and multivariate Cox analyses and Kaplan-Meier (KM) survival curves showed that patients with PI3K-Akt mutations had better overall survival (OS) than those without PI3K-Akt mutations. Genes with significant mutation rates in the two cohorts were screened by panoramic view. CIBERSORT was used to analyze changes in 22 types of immune cells to identify immune activated cells. Similarly, patients in the PI3K/Akt-mutated type (PI3K/Akt-MT) group had significantly increased immunogenicity, including increases in tumor mutation burden (TMB), neoantigen load (NAL), and MANTIS score. Using GSEA, we identified upregulated pathways related to immune response.
Conclusion: PI3K/Akt pathway mutation status can be used as an independent predictor of response to ICI treatment in COAD patients. PI3K/Akt mutations are correlated with improved OS, higher immunogenicity, greater immune response scores, and increases in activated immune cells.
Copyright © 2022 Anqi Lin et al.
Conflict of interest statement
The authors have no relevant financial or nonfinancial interests to disclose.
Figures

References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

