Theragnostic application of nanoparticle and CRISPR against food-borne multi-drug resistant pathogens
- PMID: 35711292
- PMCID: PMC9194658
- DOI: 10.1016/j.mtbio.2022.100291
Theragnostic application of nanoparticle and CRISPR against food-borne multi-drug resistant pathogens
Abstract
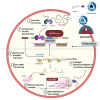
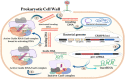
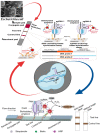
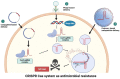
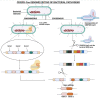
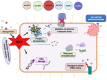
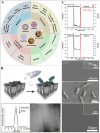
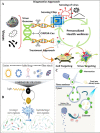
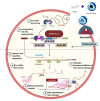
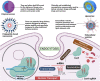
Foodborne infection is one of the leading sources of infections spreading across the world. Foodborne pathogens are recognized as multidrug-resistant (MDR) pathogens posing a significant problem in the food industry and healthy consumers resulting in enhanced economic burden, and nosocomial infections. The continued search for enhanced microbial detection tools has piqued the interest of the CRISPR-Cas system and Nanoparticles. CRISPR-Cas system is present in the bacterial genome of some prokaryotes and is repurposed as a theragnostic tool against MDR pathogens. Nanoparticles and composites have also emerged as an efficient tool in theragnostic applications against MDR pathogens. The diagnostic limitations of the CRISPR-Cas system are believed to be overcome by a synergistic combination of the nanoparticles system and CRISPR-Cas using nanoparticles as vehicles. In this review, we have discussed the diagnostic application of CRISPR-Cas technologies along with their potential usage in applications like phage resistance, phage vaccination, strain typing, genome editing, and antimicrobial. we have also elucidated the antimicrobial and detection role of nanoparticles against foodborne MDR pathogens. Moreover, the novel combinatorial approach of CRISPR-Cas and nanoparticles for their synergistic effects in pathogen clearance and drug delivery vehicles has also been discussed.
Keywords: Antimicrobial activity; CRISPR-Cas system; Diagnosis; Nanoparticle and MDR food-Borne- pathogens.
© 2022 Published by Elsevier Ltd.
Conflict of interest statement
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures

References
-
- WHO, World Health Organization WHO estimates of the global burden of foodborne diseases: foodborne disease burden epidemiology reference group 2007-2015. Encycl. Parasitol. 2015:1–265.
Publication types
LinkOut - more resources
Full Text Sources

