Bet-hedging in innate and adaptive immune systems
- PMID: 35712085
- PMCID: PMC9195227
- DOI: 10.1093/emph/eoac021
Bet-hedging in innate and adaptive immune systems
Abstract
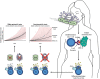
Immune system evolution is shaped by the fitness costs and trade-offs associated with mounting an immune response. Costs that arise mainly as a function of the magnitude of investment, including energetic and immunopathological costs, are well-represented in studies of immune system evolution. Less well considered, however, are the costs of immune cell plasticity and specialization. Hosts in nature encounter a large diversity of microbes and parasites that require different and sometimes conflicting immune mechanisms for defense, but it takes precious time to recognize and correctly integrate signals for an effective polarized response. In this perspective, we propose that bet-hedging can be a viable alternative to plasticity in immune cell effector function, discuss conditions under which bet-hedging is likely to be an advantageous strategy for different arms of the immune system, and present cases from both innate and adaptive immune systems that suggest bet-hedging at play.
Keywords: B cells; T cells; adaptive immunity; evolutionary medicine; immune system evolution; innate immunity; macrophages; plasticity.
© The Author(s) 2022. Published by Oxford University Press on behalf of the Foundation for Evolution, Medicine, and Public Health.
Figures
References
-
- Frank SA. Immune response to parasitic attack: evolution of a pulsed character. J Theor Biol 2002;219:281–90. - PubMed
-
- Fenton A, Lamb T, Graham AL.. Optimality analysis of Th1/Th2 immune responses during microparasite-macroparasite co-infection, with epidemiological feedbacks. Parasitology 2008;135:841–53. - PubMed
-
- Ezenwa VO, Etienne RS, Luikart G. et al. Hidden consequences of living in a wormy world: nematode‐induced immune suppression facilitates tuberculosis invasion in African buffalo. Am Nat 2010;176:613–24. - PubMed
-
- Frank SA. Specific and non-specific defense against parasitic attack. J Theor Biol 2000;202:283–304. - PubMed
-
- Metcalf CJE, Tate AT, Graham AL.. Demographically framing trade-offs between sensitivity and specificity illuminates selection on immunity. Nat Ecol Evol 2017;1:1766–72. - PubMed
LinkOut - more resources
Full Text Sources

