TMBIM5 loss of function alters mitochondrial matrix ion homeostasis and causes a skeletal myopathy
- PMID: 35715207
- PMCID: PMC9206080
- DOI: 10.26508/lsa.202201478
TMBIM5 loss of function alters mitochondrial matrix ion homeostasis and causes a skeletal myopathy
Abstract
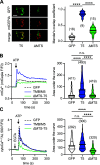
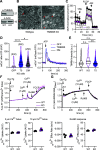
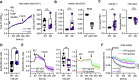
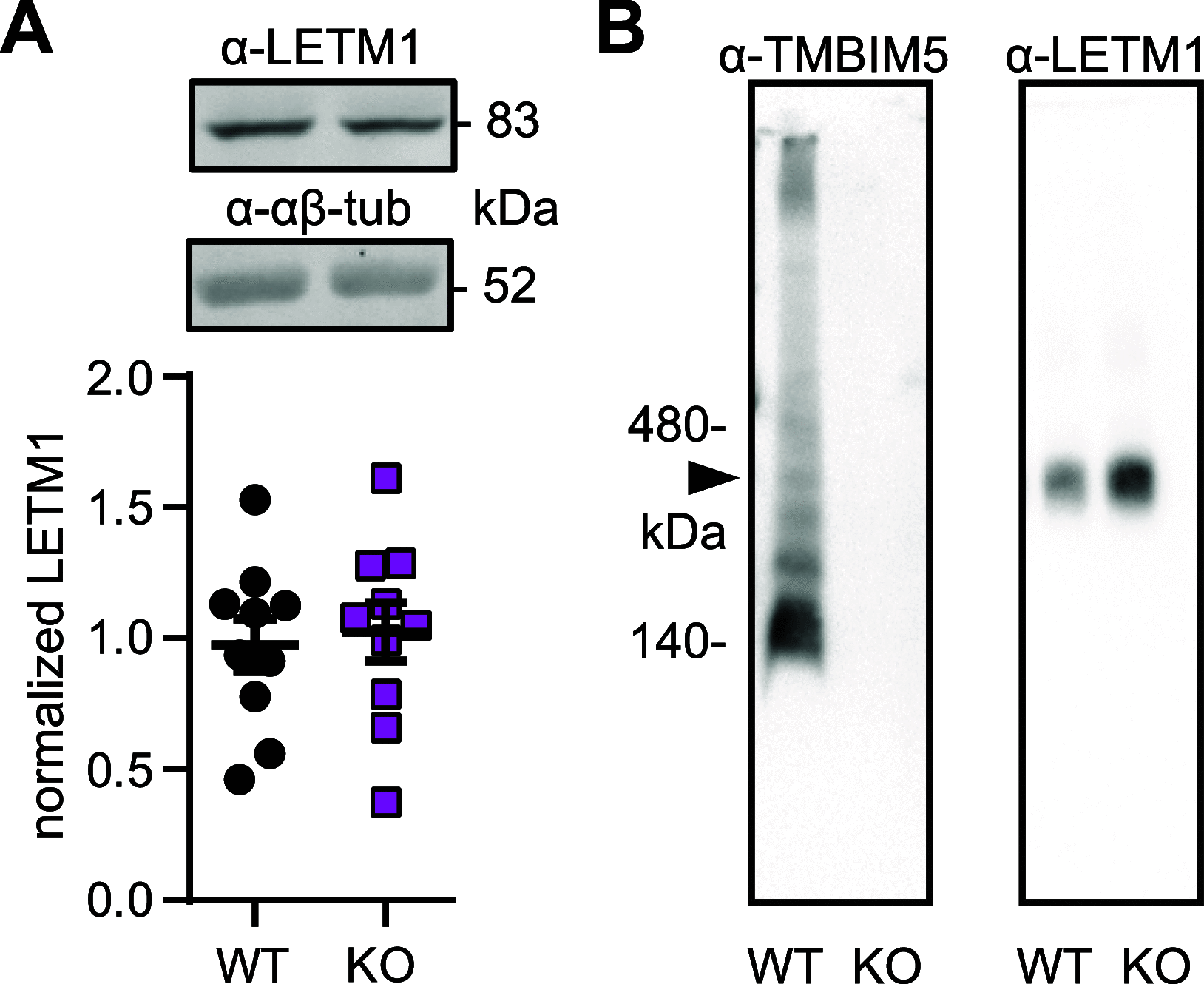
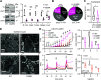
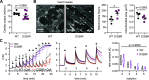
Ion fluxes across the inner mitochondrial membrane control mitochondrial volume, energy production, and apoptosis. TMBIM5, a highly conserved protein with homology to putative pH-dependent ion channels, is involved in the maintenance of mitochondrial cristae architecture, ATP production, and apoptosis. Here, we demonstrate that overexpressed TMBIM5 can mediate mitochondrial calcium uptake. Under steady-state conditions, loss of TMBIM5 results in increased potassium and reduced proton levels in the mitochondrial matrix caused by attenuated exchange of these ions. To identify the in vivo consequences of TMBIM5 dysfunction, we generated mice carrying a mutation in the channel pore. These mutant mice display increased embryonic or perinatal lethality and a skeletal myopathy which strongly correlates with tissue-specific disruption of cristae architecture, early opening of the mitochondrial permeability transition pore, reduced calcium uptake capability, and mitochondrial swelling. Our results demonstrate that TMBIM5 is an essential and important part of the mitochondrial ion transport system machinery with particular importance for embryonic development and muscle function.
© 2022 Zhang et al.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures


Similar articles
-
TMBIM5 is the Ca2+ /H+ antiporter of mammalian mitochondria.EMBO Rep. 2022 Dec 6;23(12):e54978. doi: 10.15252/embr.202254978. Epub 2022 Nov 2. EMBO Rep. 2022. PMID: 36321428 Free PMC article.
-
Transmembrane BAX Inhibitor-1 Motif Containing Protein 5 (TMBIM5) Sustains Mitochondrial Structure, Shape, and Function by Impacting the Mitochondrial Protein Synthesis Machinery.Cells. 2020 Sep 23;9(10):2147. doi: 10.3390/cells9102147. Cells. 2020. PMID: 32977469 Free PMC article.
-
Deletion of mitochondrial calcium uniporter incompletely inhibits calcium uptake and induction of the permeability transition pore in brain mitochondria.J Biol Chem. 2018 Oct 5;293(40):15652-15663. doi: 10.1074/jbc.RA118.002926. Epub 2018 Aug 28. J Biol Chem. 2018. PMID: 30154242 Free PMC article.
-
The Mitochondrial Permeability Transition Pore-Current Knowledge of Its Structure, Function, and Regulation, and Optimized Methods for Evaluating Its Functional State.Cells. 2023 Apr 27;12(9):1273. doi: 10.3390/cells12091273. Cells. 2023. PMID: 37174672 Free PMC article. Review.
-
Mitochondrial ion channels as oncological targets.Oncogene. 2014 Dec 4;33(49):5569-81. doi: 10.1038/onc.2013.578. Epub 2014 Jan 27. Oncogene. 2014. PMID: 24469031 Review.
Cited by
-
Post-translational modifications and protein quality control of mitochondrial channels and transporters.Front Cell Dev Biol. 2023 Aug 3;11:1196466. doi: 10.3389/fcell.2023.1196466. eCollection 2023. Front Cell Dev Biol. 2023. PMID: 37601094 Free PMC article. Review.
-
The roles of mitochondria in global and local intracellular calcium signalling.Nat Rev Mol Cell Biol. 2025 Jun;26(6):456-475. doi: 10.1038/s41580-024-00820-1. Epub 2025 Jan 27. Nat Rev Mol Cell Biol. 2025. PMID: 39870977 Review.
-
TMBIM5 is the Ca2+ /H+ antiporter of mammalian mitochondria.EMBO Rep. 2022 Dec 6;23(12):e54978. doi: 10.15252/embr.202254978. Epub 2022 Nov 2. EMBO Rep. 2022. PMID: 36321428 Free PMC article.
-
Mitochondrial Cation Signalling in the Control of Inflammatory Processes.Int J Mol Sci. 2023 Nov 24;24(23):16724. doi: 10.3390/ijms242316724. Int J Mol Sci. 2023. PMID: 38069047 Free PMC article. Review.
-
Simultaneous Measurement of Changes in Mitochondrial and Endoplasmic Reticulum Free Calcium in Pancreatic Beta Cells.Biosensors (Basel). 2023 Mar 14;13(3):382. doi: 10.3390/bios13030382. Biosensors (Basel). 2023. PMID: 36979594 Free PMC article.
References
-
- Baughman JM, Perocchi F, Girgis HS, Plovanich M, Belcher-Timme CA, Sancak Y, Bao XR, Strittmatter L, Goldberger O, Bogorad RL, et al. (2011) Integrative genomics identifies MCU as an essential component of the mitochondrial calcium uniporter. Nature 476: 341–345. 10.1038/nature10234 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
